Ready For Sale
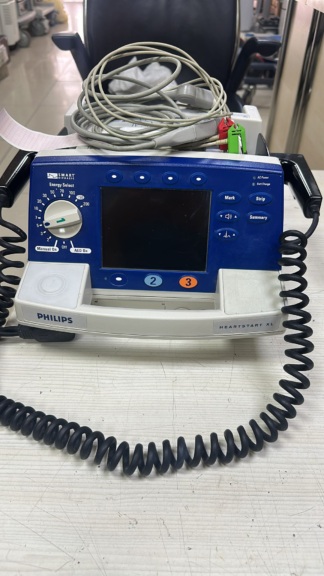
Secondhand Medtronic Lifepak CR Plus AED Defibrillator
Price: USD$ 1.250,00 Approx: 56.250,00 TL
Ready For Sale
Ask a Question
Payment
No additional fees, full assurance. We provide complete financial and operational security in secondhand medical device trading. For this, we offer the "Secure Payment" service. This free service protects the rights of both parties by securing the buyer's money and the seller's product. The Secure Payment system is a standard assurance mechanism offered by Medbidding. For additional information, review the "services" page.
There is no cash on delivery order system on the Medbidding platform. For payments to be made by credit card, the product to be purchased must comply with this payment method. You can contact us to get information about this. We would be happy to assist you.
For payments made outside of Turkiye, you can choose bank transfer, credit card, Western Union or cryptocurrency options. Installment options are not currently available for credit cards other than Turkish banks.
Shipping
Standard Shipping Conditions
In order to ensure secure transactions on Medbidding, the shipping process is managed through four different scenarios depending on the location of the buyer and seller. Free shipping is available for some categories. The terms below apply to all categories unless otherwise stated.
Buyer and Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer Outside Turkiye, Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer in Turkiye, Seller Outside Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer and Seller Outside Turkiye
If there is a local operation center in the seller's country:
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
If there is no operation center in the seller's country:
- Technical Inspection: Inspections are performed by our engineers via remote video call.
- Seller → Buyer: The seller packages the product and sends it directly to the buyer's address. The seller is responsible for this shipping cost.
Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 04.03.2026
PHYSIO-CONTROL LIFEPAK CR Plus Defibrillator Analysis Report
Device Identification, Brand and Model
The device shown in the visuals is an Automatic External Defibrillator (AED) used in medical emergencies. Brand and model information has been definitively identified as a result of examinations on the device’s front panel and the label on its carrying case.
- Brand: PHYSIO-CONTROL
- Model: LIFEPAK CR Plus
- Product Type: Defibrillator (Automatic/Semi-Automatic)
Originality and General Condition
The “PHYSIO-CONTROL” logos, font styles, color scheme (yellow/black/grey combination), and design details of the carrying case on the product indicate that it belongs to the original manufacturer. No inconsistency suggesting it is an aftermarket or replica product has been detected.
The device is in used condition overall. There is dirt on its surface due to storage or field use conditions. Rather than appearing “new” or “refurbished,” it has the appearance of a device with an active use history.
Areas of Use
The PHYSIO-CONTROL LIFEPAK CR Plus is designed to analyze heart rhythm and apply a shock when necessary in patients experiencing sudden cardiac arrest. It operates on a semi-automatic or fully automatic principle. Areas of use include:
- Pre-hospital emergency care (Ambulances, paramedics).
- Public areas (Airports, shopping malls, gyms).
- Workplace health and safety units.
- Family practices and clinics.
Quantity Information
The following components are visible in the visuals:
- 1 LIFEPAK CR Plus Defibrillator Unit
- 1 Original LIFEPAK Carrying Case (Black, soft cover)
Physical Deformation and Cosmetic Condition
The following details were observed during visual inspection of the device and case:
- Front Panel: The device’s dark grey front panel exhibits widespread dust accumulation and particulate matter. A need for cleaning has been observed.
- Protective Covering: In the first visual, a transparent material (tape residue, protective gelatin, or a loose plastic piece) extending diagonally appears to be independent of the device’s transparent cover, located above the “LIFEPAK CR Plus” inscription. The surface of this area does not appear smooth.
- Case Structure: No cracks or deep dents are discernible on the yellow plastic case in the visual, but surface dirt is prominent.
- Case Label: The white label on the case shows wear due to friction, dirt stains, and slight fraying at the corner seams. The “LifePak Defibrillator” inscription is legible.
Mechanical and Electronic Component Analysis
- Lid Mechanism: A large white arrow indicating the opening direction is present on the device. The lid latch appears to be in a fully closed position in the visual.
- Buttons: A yellow “ON/OFF” button is present on the front panel. Button integrity is preserved.
- Display Panel (LCD): A small LCD status indicator is located directly below the device’s upper carrying handle, on the upper part of the lid. In the visual, faint black segments resembling a battery status icon are barely discernible on this screen. There is no indication of the screen being physically broken.
Accessories and Battery Status
- Case: The product is housed within its original black textile carrying case. The case features an embossed heart and lightning bolt logo on top, along with a rubber “LIFEPAK” inscribed carrying handle.
- Battery (CHARGE-PAK): These models typically use a replaceable CHARGE-PAK charging stick. Since the battery compartment is closed, the physical condition or expiration date of the battery cannot be read from the visual.
- Electrodes (Pads): As the device is closed, the presence, type (adult/pediatric), or expiration dates of the electrodes cannot be confirmed.
Potential Risk of Malfunction
The cosmetic condition of the device suggests it has not been maintained for some time or has been stored in a dusty environment. Observed potential risk factors include:
- The dirt on the front panel and the irregularity in the transparent area indicate that the device needs hygienic cleaning.
- Since the full charge level of the battery on the LCD screen cannot be clearly confirmed from the visual, the possibility of the battery being depleted or having reached the end of its life is common for such stored devices. Replacement of the power supply unit (CHARGE-PAK) may be necessary.
- The shelf life of the device’s internal electrodes (pads) could not be checked from the visual; their expiration date may have passed.
Conclusion and Recommendation
The PHYSIO-CONTROL LIFEPAK CR Plus defibrillator in the visual is a used device that maintains its physical integrity but requires cosmetic cleaning and technical maintenance. No breaks, missing parts, or severe impact marks have been found. Before the device is put into use, it is essential to have a calibration check performed by an authorized biomedical service, inspect the battery and electrode expiration dates, and verify whether the “Self-Test” function reports “OK”.