Ready For Sale
Secondhand Armoline ALC-411C 1 Channel ECG Device
Price: USD$ 180,00 Approx: 8.100,00 TL
Ready For Sale
Ask a Question
Payment
No additional fees, full assurance. We provide complete financial and operational security in secondhand medical device trading. For this, we offer the "Secure Payment" service. This free service protects the rights of both parties by securing the buyer's money and the seller's product. The Secure Payment system is a standard assurance mechanism offered by Medbidding. For additional information, review the "services" page.
There is no cash on delivery order system on the Medbidding platform. For payments to be made by credit card, the product to be purchased must comply with this payment method. You can contact us to get information about this. We would be happy to assist you.
For payments made outside of Turkiye, you can choose bank transfer, credit card, Western Union or cryptocurrency options. Installment options are not currently available for credit cards other than Turkish banks.
Shipping
Free Shipping Conditions
You can benefit from our FREE SHIPPING campaign for your purchases in this category. Medbidding covers all shipping costs for both the buyer and the seller on orders that meet the conditions below. For all international sales and domestic sales within Turkiye that do not meet these conditions, Medbidding’s standard shipping procedures will apply.
Conditions
- The product must be sold for $350 USD or more.
- Both the buyer and the seller must be located in Turkiye.
Standard Shipping Conditions
In order to ensure secure transactions on Medbidding, the shipping process is managed through four different scenarios depending on the location of the buyer and seller. Free shipping is available for some categories. The terms below apply to all categories unless otherwise stated.
Buyer and Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer Outside Turkiye, Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer in Turkiye, Seller Outside Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer and Seller Outside Turkiye
If there is a local operation center in the seller's country:
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
If there is no operation center in the seller's country:
- Technical Inspection: Inspections are performed by our engineers via remote video call.
- Seller → Buyer: The seller packages the product and sends it directly to the buyer's address. The seller is responsible for this shipping cost.
Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 31.03.2026
Report code: 1774956934
ArmoLine ECG Device Analysis Report
Device Identification and Areas of Use
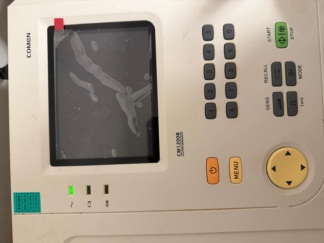
The product in the images is an Electrocardiography (ECG) device used to measure, monitor, and record patients’ heart rhythm and electrical activity. It is suitable for use in cardiology departments, emergency rooms, clinics, and general practices. The brand of the device has been identified as ArmoLine from the logo located in the upper right corner of the device. No label or writing indicating the device’s model is present in the images.
General Condition and State
The general condition of the device is very high. The presence of the factory-applied transparent protective film with a blue frame on the screen indicates that the device is new or has been used very little. No yellowing, discoloration, dirt, or dust accumulation has been observed on the outer plastic casing.
Physical Deformation and Mechanical Components
There are no scratches, cracks, breaks, or dents on the device’s body. No fading, bubbling, or wear is present on the membrane keypad on the front panel. The integrated thermal printer cover on the left side is closed and intact. The physical integrity of the buttons and mechanical parts has been preserved.
Electronic Components and Screen Analysis
It has been confirmed in the fourth image that power was supplied to the device by plugging a cable into the power socket on the right side of the device, and that the device is electronically operational. The two green LED indicators above the power button are lit. The LCD screen has illuminated without issues, and the interface has loaded. No dead pixels or darkening are visible on the screen. The following data can be read on the screen interface:
- 1CH (1 Channel) indication in the upper left corner
- Time information in the upper right corner
- List of standard 12-lead ECG channels (I, II, III, aVR, aVL, aVF, V1, V2, V3, V4, V5, V6)
- AUTO, 10, 25, HUM, EMG, ADS parameters indicating acquisition mode and filter settings in the footer bar
Accessories and Quantity Information
The device’s original cardboard box and custom-cut white foam supports that protect the device against impacts are present. No missing parts are visible in the box contents. The hardware and accessories identified in the images are:
- 1 ArmoLine ECG main unit
- 1 ECG patient cable with multiple leads
- 4 blue clamp-type limb electrodes
- 6 metal-headed suction-bulb type chest electrodes
- 1 yellow-green grounding cable
- 1 black power cable
- 1 small white box with a green QC PASS label
- 1 user manual or file partially visible under the cables
Warnings and Label Information
Two safety warnings are printed in English on the lower left corner of the device’s front panel. The first warning states that use in environments containing flammable anesthetic agents may create an explosion hazard. The second warning emphasizes that for continued protection against fire hazard, only fuses of the same type and rating should be used when replacing fuses. Since the rear or bottom surface of the device is not visible in the images, specific label information such as serial number, manufacturing year, lot number, or REF code could not be accessed.
Potential Malfunction Risk
Based on the visual data, no malfunction or damage has been detected in the device or its accessories. There are no crushes, breaks, or rust in the cables or sockets. The device’s screen and power system are actively working. The product’s condition is excellent, and it does not carry any potential malfunction risk in the visual context.