Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 07.10.2025
Petas Kardiopet 300 Medical Device Analysis Report
Overview and Product Identification
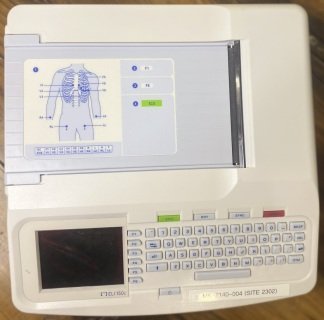
The device shown in the visuals is a Petas Kardiopet 300 model, according to the brand and model information on it. The inscription “EKG GİRİŞİ” (ECG INPUT) on the front panel of the device and its general structure indicate that the product is a medical electrocardiography (ECG) device. The visual shows 1 main unit and a cable set connected to this unit, with electrode-like parts at their ends. The overall appearance of the device indicates that it is an old model.
Physical Condition and State Analysis
The device generally appears heavily used and neglected for a long time. There is a significant amount of dust and dirt on its casing and accessories. This situation suggests that the product has not been actively used and has been stored in a warehouse. No packaging or box contents are present in the visual.
- Casing: Significant yellowing and discoloration are observed on the device’s cream-colored plastic casing. There is general dirtiness on the surface. No distinct cracks, breaks, or dents were detected on the casing in the visuals.
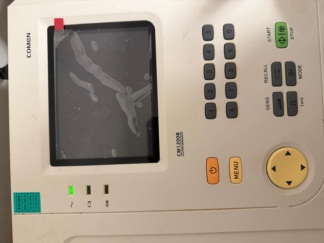
- Screens: The device has two screens. The screens are off and dusty. It cannot be determined whether they are functional or if there are any display defects (scratches, stains, pixel errors).
- Cables and Accessories: The cable set connected to the device is quite tangled and dusty. No visible breaks, compressions, or peeling are present in the cable insulation. A red clip (electrode clip), which is part of the set, can be discerned.
Technical and Mechanical Evaluation
The mechanical and visible technical parts of the device are generally in place. Control buttons on the front panel with functions such as “EKG” (ECG), “YARDIM” (HELP), and “KAYIT” (RECORD) appear physically intact. There is an adjustment knob on the right side of the device and a metal “EKG GİRİŞİ” (ECG INPUT) socket on the front panel. Since the device is turned off, no comment can be made regarding the functionality of the electronic components.
Label and Model Information
Based on the examinations in the visuals, the only readable information on the device is the brand and model.
- Brand: Petas
- Model: Kardiopet 300
Any label containing details such as serial number, REF code, lot number, or year of manufacture on the product is either unreadable due to visual angles or is not present.
Potential Failure Risks and Recommendations
The device’s age, the prominent yellowing of its casing, and the accumulated dense dust indicate that it has not been used for a long time and has likely been neglected. This situation carries the potential to create age-related performance issues in internal electronic components. Furthermore, it should be considered that the cables may have lost their flexibility over time. Due to the device’s current condition, it is strongly recommended that it be checked, cleaned, and calibrated by an authorized technical service before any use.