Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 31.10.2025
Comen CM1200B Electrocardiograph Device Analysis Report
Overview and Product Description
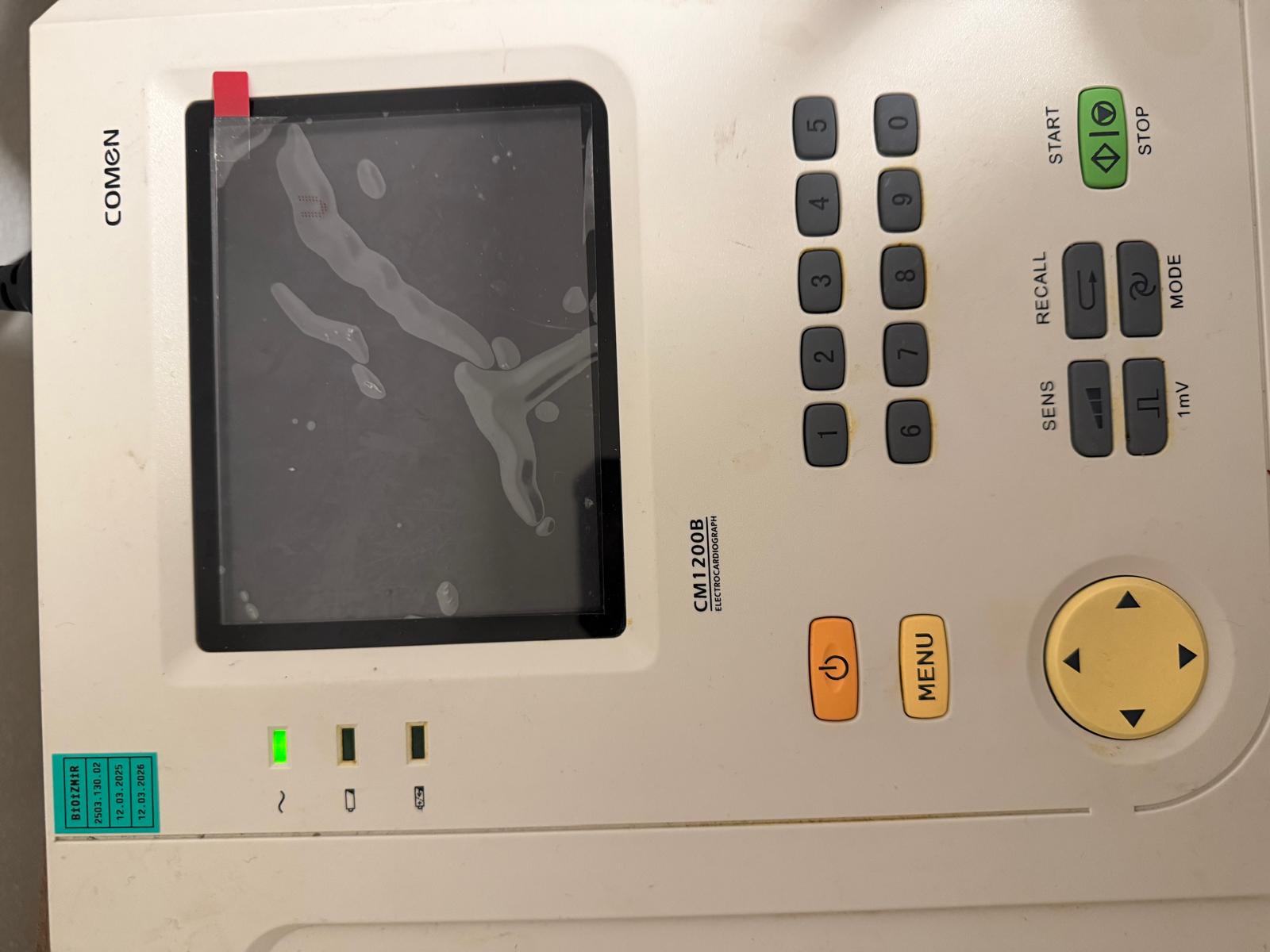
The product shown in the images is a Comen brand CM1200B model electrocardiograph (ECG) device. This device is a medical equipment used in healthcare institutions to measure and record the electrical activity of the heart. Only the main unit of the device is visible in the image, and no accessories are present. The general appearance of the product indicates that it has been previously used.
Brand and Model Identification
During a detailed visual inspection, the “COMEN” brand logo is clearly visible in the upper left corner of the device, and the model and device type information “CM1200B ELECTROCARDIOGRAPH” is clearly legible below the screen. This information conclusively confirms that the product’s brand is Comen and its model is CM1200B. The quality of the text and logo on the device suggests that the product is original.
Physical Condition and Deformation Assessment
The general condition of the device is assessed as used. Observations regarding its physical condition are detailed below:
- Casing: No significant cracks or fractures were observed on the device’s white plastic casing. However, dirt, stains, and light scratches are present in places, particularly around the keypad and on its general surface.
- Screen: A protective film is present on the device’s screen. This protective film is quite worn; it has numerous air bubbles and has started to lift at the edges. Due to the condition of this protective film, the state of the actual glass surface underneath the screen cannot be clearly assessed.
- Keypad: All buttons are physically in place and appear intact. Dirt accumulation, believed to be due to usage, is noticeable around the buttons. Functional labels such as “START/STOP”, “RECALL”, “SENS”, “MODE”, “MENU” are present on the buttons.
Technical and Operational Observations
The image shows some indications that the device is plugged in and operational.
- Power Status: The power indicator light (~) located on the lower left side of the device is illuminated green. This indicates that the device is connected to an alternating current (AC) power source and is powered on.
- Other Indicators: Next to the power light, there are icons and corresponding LED slots indicating battery and SD card status, but these lights are not active in the image.
- Electronic Components: As far as visible, no external electronic problems (such as burns, damaged connectors, etc.) have been detected. A black power cable connected to the device is visible on the left side.
Label Information and Calibration
A green label is located on the lower left corner of the device. The information on this label is as follows:
- Label Title: BİOZİMR
- Code: 2503.130.02
- Dates: The label includes the dates “12.03.2025” and “12.03.2026”. These dates are presumed to be the device’s calibration validity periods or maintenance dates.
Accessories
No accessories (such as ECG cables, electrodes, user manual, carrying case, etc.) are present in the image apart from the device’s main unit. Therefore, it is understood that the device is missing accessories.
Conclusion and Potential Risks
The Comen CM1200B ECG device in the images is an operational product showing visible signs of use. While there is no major damage to the device’s casing, it clearly requires general cleaning and maintenance. The most significant potential risk is the unknown true condition of the screen underneath the protective film. The extensive wear on the protective film may indicate that the screen itself is similarly worn, but this is only a possibility. Although the device is believed to perform its basic functions, it needs to be tested for a full performance evaluation, and missing accessories such as ECG cables need to be procured.