Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 27.11.2025
Nihon Kohden Cardiofax Q ECG-9132K ECG Device Analysis Report
Device Identification
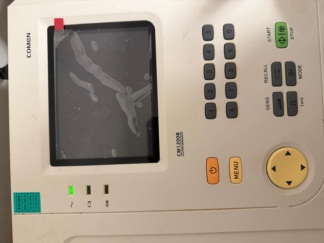
The image displays a medical diagnostic device. The device is a portable ECG (Electrocardiography) unit with an integrated screen, an alphanumeric keypad, and function keys. The product has a design that allows it to be mounted on medical trolleys or stands.
Brand and Model
Upon examining the inscriptions and labels on the device, the brand and model information is clearly legible:
- Brand: NIHON KOHDEN
- Series/Product Group: cardiofax Q
- Model: ECG-9132K (Clearly stated on the bottom right corner of the control panel).
Based on this data, the product has been identified with 100% certainty as a professional ECG device used in hospitals and clinics to monitor and record heart rhythm.
Originality
The device’s logo placement, font usage, keypad layout, and material quality are consistent with Nihon Kohden’s standard design language. No signs of imitation, misprints, or poor craftsmanship are apparent. The product is assessed to be an original medical device.
Areas of Use
This device is designed for use in cardiology outpatient clinics, emergency rooms, intensive care units, and general medical practices. Its primary function is to record, monitor, and report patients’ cardiac electrical activities (ECG). The device in the image is an ECG monitor and recorder; it collects data via electrodes (leads) placed on the patient.
Quantity Information
Only 1 main device (Nihon Kohden ECG unit) is visible in the image. Only a portion of the metal stand/trolley on which the device is mounted or placed is visible, but the stand is a separate unit from the device.
General Condition and Physical State
The device generally maintains its integrity, and no major fragmentation is visible on its casing. However, evident cosmetic signs indicate that the device has been used. It has been observed that the device is not “new” but a second-hand product actively used in the field.
Physical Deformation and Cosmetic Examination
- Screen: The screen has significant dust accumulation and light stains. No deep scratches or cracks were detected on the screen glass in the image, but the surface requires cleaning.
- Casing: The device’s outer plastic casing is beige/light grey. Slight color changes on plastic surfaces due to aging are normal. A small black screw or connection point is visible on the bottom right side, near the stand.
- Keypad: The prints (letters and symbols) on the keys are legible. No excessive wear is observed on the keys.
Mechanical and Electronic Components
- Keys: The device features F1, F2, F3 function keys, a full QWERTY character set, a numerical keypad, directional keys, and a prominent green “Start/Stop” button. The physical structure of the keys appears robust. No missing keys.
- Input/Output Ports: Patient cable (ECG lead) inputs are not visible on the device’s top surface; it should be noted that in this model, inputs are typically located on the side or rear panel, but they are not visible in this image.
- Status LEDs: In the upper left corner, there are indicator icons for AC power, Battery charge status, and Paper/Printer status. Since the device is powered off, the operational status of these LEDs could not be analyzed.
Accessories
No ECG cable (lead), power cable, or medical sensors are attached to the device in the image. Only the main body of the device is visible. The metal surface underneath might belong to a trolley, but the overall trolley and its condition cannot be analyzed from this perspective.
Battery Status
The battery icon on the control panel indicates that the device has an internal battery or supports this option. However, whether the battery is installed or its capacity cannot be determined from the image.
Screen Analysis
The device’s screen is off (black). The screen is an LCD panel. Since there is no display on the screen, no comments can be made regarding pixel defects or backlight status.
Label Information
Main label and model information located on the front panel:
- Brand: NIHON KOHDEN
- Series Name: cardiofax Q
- Model Code: ECG-9132K
Potential Fault Risk
Although the device’s exterior appears robust, the dust accumulation on the screen surface suggests that the device might have been stored for some time. Potential risks include:
- The possibility of the internal battery having reached the end of its life due to the device not being used for a long period.
- The possibility of fine hairline scratches appearing underneath when the screen surface dirt is cleaned.
- The possibility of paper jamming or print head issues, as the condition of the thermal printer mechanism (its cover is located below the screen) could not be determined from the image.
Overall, no physical breakage or heavy damage is observed, and the cosmetic condition is moderate-to-good.