Ready For Sale
Secondhand Comen CM100 1 Channel ECG Device
Price: USD$ 260,00 Approx: 11.700,00 TL
Ready For Sale
Ask a Question
Payment
No additional fees, full assurance. We provide complete financial and operational security in secondhand medical device trading. For this, we offer the "Secure Payment" service. This free service protects the rights of both parties by securing the buyer's money and the seller's product. The Secure Payment system is a standard assurance mechanism offered by Medbidding. For additional information, review the "services" page.
There is no cash on delivery order system on the Medbidding platform. For payments to be made by credit card, the product to be purchased must comply with this payment method. You can contact us to get information about this. We would be happy to assist you.
For payments made outside of Turkiye, you can choose bank transfer, credit card, Western Union or cryptocurrency options. Installment options are not currently available for credit cards other than Turkish banks.
Shipping
Free Shipping Conditions
You can benefit from our FREE SHIPPING campaign for your purchases in this category. Medbidding covers all shipping costs for both the buyer and the seller on orders that meet the conditions below. For all international sales and domestic sales within Turkiye that do not meet these conditions, Medbidding’s standard shipping procedures will apply.
Conditions
- The product must be sold for $350 USD or more.
- Both the buyer and the seller must be located in Turkiye.
Standard Shipping Conditions
In order to ensure secure transactions on Medbidding, the shipping process is managed through four different scenarios depending on the location of the buyer and seller. Free shipping is available for some categories. The terms below apply to all categories unless otherwise stated.
Buyer and Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer Outside Turkiye, Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer in Turkiye, Seller Outside Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer and Seller Outside Turkiye
If there is a local operation center in the seller's country:
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
If there is no operation center in the seller's country:
- Technical Inspection: Inspections are performed by our engineers via remote video call.
- Seller → Buyer: The seller packages the product and sends it directly to the buyer's address. The seller is responsible for this shipping cost.
Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 21.11.2025
Comen CM 100 Electrocardiograph Device Analysis Report
Device Identification, Brand and Model
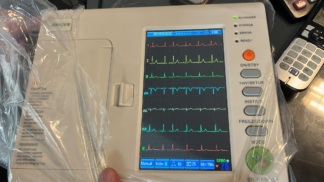
The device in the analyzed visuals is a portable Electrocardiograph (ECG) device used for medical diagnostic purposes. Upon examining the type labels on the front face’s bottom left corner and the rear face of the product, its brand has been definitively identified as COMEN and its model as CM 100 with 100% certainty. The phrase “Electrocardiograph” is clearly visible on both the front face and the rear label of the device.
Areas of Use
The COMEN CM 100 is a professional medical device used to record, monitor, and report the electrical activities of patients’ hearts (ECG). The accessories in the visuals indicate that the device is equipped for standard ECG recordings.
General Condition and Originality
The device is visually clean and in a preserved condition. No yellowing or significant discoloration has been observed on the plastic casing. The manufacturer information, serial number format, and workmanship quality on the rear label indicate that the product is an original COMEN item. The presence of a calibration label on the front handle of the device suggests that the product has been previously used in a professional setting (second-hand) but is well-maintained.
Physical Deformation and Cosmetic Examination
Upon examining the product’s outer casing:
- No deep scratches, cracks, breaks, or dents have been detected on the front panel and body.
- The joints of the plastic components appear regular.
- While no deformation preventing use is visible on the screen surface, no comment can be made regarding pixel status as the device is turned off.
- The rubber feet on the bottom of the device are complete and intact, with no missing parts observed.
Mechanical and Electronic Components
Mechanical and electronic details based on visual inspection are as follows:
- Keypad: The keypad, observed to be of membrane type (On/Off, Menu, Print/Stop, arrow keys, etc.), appears physically sound. No tears or collapses have been observed.
- Printer Section: The thermal printer cover located on the right side is in place, and its mechanical integrity appears solid.
- Ventilation: The ventilation grilles on the back of the device are clean, with minimal dust accumulation.
- Connection Ports: The power input and grounding screw located at the rear are physically undamaged.
Accessories and Box Contents
The accessories presented with the device in the visuals and found within the box are as follows:
- 1 unit COMEN CM 100 ECG Main Unit.
- 1 set ECG patient cable (Lead wire).
- 4 Limb Clamps (Blue, extremity electrodes).
- 6 Chest Electrodes (Vacuum bulb type, grey).
- 1 black power cable (Schuko plug type).
- 1 grounding cable (Yellow-green, visible in the transparent bagged box compartment).
- 1 roll of thermal printer paper (For spare or use).
- User manual or documentation (Documents with “COMEN” logo are visible under the cables).
Label Information and Technical Data
Technical data and identification information obtained from the label on the rear side of the device are as follows:
- Model: CM 100
- Manufacturer: Shenzhen Comen Medical Instruments Co., Ltd.
- Serial Number (SN): 10150512019A
- Manufacturing Date: 2015.05.26 (May 26, 2015)
- Power Input: 220V~, 50Hz/60Hz
- Power Consumption: 35VA
- European Representative (EC REP): Lotus Medical Equipment Limited (Ireland)
Furthermore, a green label belonging to the company “ERENLER KALİBRASYON” is located on the front handle. This label displays the code “ERN-5057”, with the Calibration Date (CD) specified as 21.01.2016 and the Validity Date (VD) as 21.01.2017. This information proves that the device was actively used in the past and underwent periodic checks.
Battery Status
There is a separate cover and label for the battery on the rear of the device. According to the information in the visual:
- Type: Rechargeable Lithium Battery (Lithium Battery Rack).
- Voltage: 14.4V
- Capacity: 1600mAh
Although it cannot be determined from the visuals whether the battery is physically swollen, the battery cover is properly closed. The current charge retention capacity of the battery cannot be included in the visual analysis.
Screen and Interface Analysis
The device features an LCD screen, estimated to be monochrome. Since the device was photographed in an off (unpowered) state, no warnings, error codes, or menu displays are visible on the screen. LED status indicators (Mains, Battery, etc.) are present next to the screen.
Potential Malfunction Risk and Conclusion
Based on visual inspections, no serious potential malfunction risk such as broken parts, corrosion (rusting), cable stripping, or traces of liquid contact detectable externally has been observed on the device. However, due to the product being manufactured in 2015, there is a possibility that its internal battery may have reached the end of its life. Furthermore, considering that the calibration date expired in 2017, a new calibration and functional test may be required before professional use. The device and its accessories appear clean, well-maintained, and complete in terms of overall condition.