Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 02.02.2026
BiO2Flow High Flow Oxygen Therapy Device Analysis Report
Device Identification
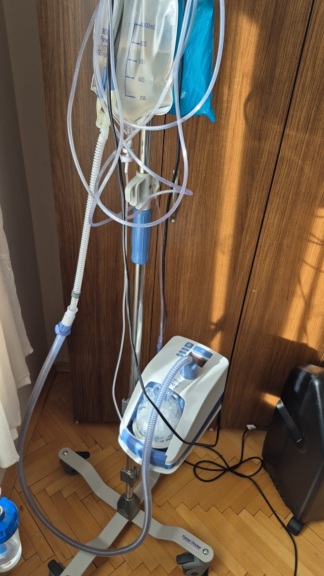
The device shown in the visuals is a High Flow Oxygen Therapy device manufactured for medical use. The device is a mobile unit designed to provide heated and humidified oxygen at a high flow rate to patients experiencing respiratory distress. The unit is mounted on a wheeled stand and is suitable for bedside use.
Brand and Model
As a result of detailed analysis, the brand of the device has been identified as BiO2Flow (Biosys). The “biO2flow” logo is clearly visible on the front face of the product, in the lower right corner. The model name is also referred to by the same name. The presence of Turkish warnings and menus on the device labels (such as “Sıcaklık” – Temperature, “Isıtıcı” – Heater) confirms that the product is intended for the Turkish market or is a locally produced device (manufactured by Biosys).
Authenticity
Upon examination of the product casing, screen integrity, label quality, workmanship at the connection points (color-coded ports), and logo placement, the product is understood to be Original. The professional printing of port labels like “Sıcaklık” (Temperature) and “Isıtıcı” (Heater) onto the device casing indicates that they are not aftermarket additions.
Areas of Use
This device is used in intensive care units (ICU), pulmonology departments, and emergency departments for adult patients experiencing respiratory failure, low blood oxygen levels, or requiring respiratory support (but not intubated). It delivers a humidified and heated air/oxygen mixture to the patient via a high-flow nasal cannula.
Overall Condition and Cosmetic State
Although the device generally appears clean and ready for use, it is a used product.
- Physical Deformation: No cracks, deep scratches, or broken parts have been observed on the device casing. The plastic components are intact and have retained their color.
- Contamination: No significant dirt or dust accumulation is visible on the device. Water is present in the humidifier reservoir, indicating that the device has been recently tested or used.
- Stand Condition: The device is on its original wheeled stand, and the stand appears sturdy.
Mechanical and Electronic Components
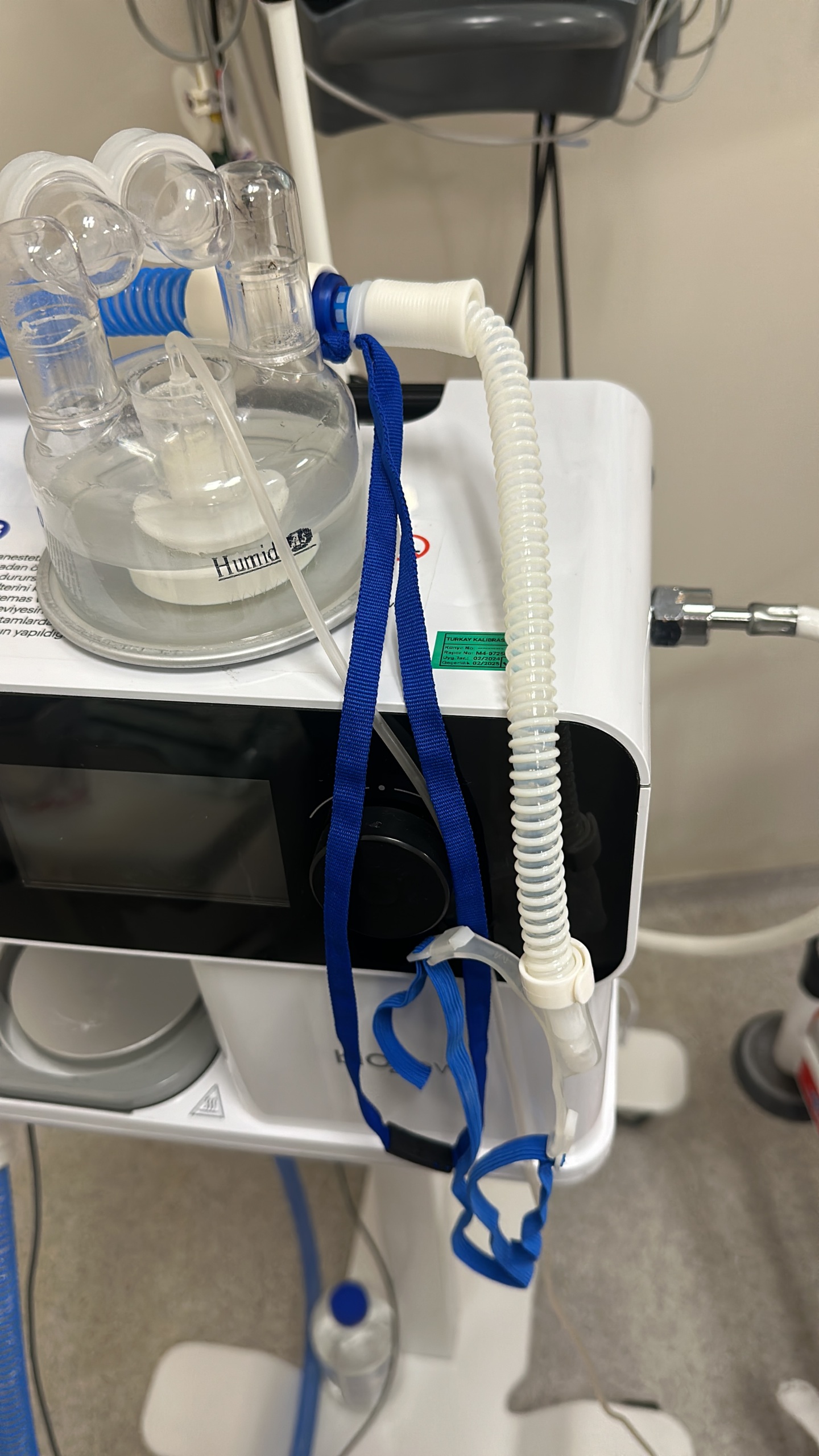
- Ports and Connections: The “Inspiration” (Air outlet), “Heater Wire” (Heater cable – Yellow Port), and “Temp. Probe” (Temperature probe – Blue Port) sockets on the right side panel of the device are intact. No bending or rusting of the pins has been visually detected.
- Nebulizer Port: A “Nebulizer” labeled outlet is present on the side panel.
- Screen: The LCD screen is functional; no dead pixels or cracks have been detected, but a critical warning is present on the screen (Details are under the “Existing Fault” heading).
- Humidifier Reservoir: On the left side of the device, there is a transparent humidifier water reservoir placed on the heating base. Water is present in the reservoir.
Existing Fault
During the screen analysis in Visual 3, a significant fault notification has been detected. The red warning strip at the top of the device screen displays “HW Error” (Hardware Error).
This error indicates a technical problem in one of the device’s hardware components (such as sensors, motherboard, or power supply). In this state, the device may not be suitable for patient use and may require technical service intervention.
Screen Analysis and Operating Parameters
The screen data at the time of photography are as follows:
- Warning Message: HW Error (On a red background).
- FLOW RATE: Set or measured as 50 LPM (50 Liters Per Minute).
- O2 RATE: At 100% O2 (Pure oxygen) level.
- Firmware Version: The software version “rfs0.0.46 ui2.4.2” is visible at the bottom of the screen.
Accessories
The accessories identified in the visual are as follows:
- 1 BiO2Flow Device Main Unit.
- 1 Wheeled Transport Stand.
- 1 Humidifier Water Reservoir (Attached to the device, brand not fully legible but a standard compatible reservoir).
- 1 White Corrugated Respiratory Tube/Circuit.
- 1 Circuit carrying/fixing arm (Blue striped arm).
- Note: The heater cable and temperature probe sockets appear empty; cables are not connected.
Label and Calibration Information
A green “TURKAY CALIBRATION” label is located on the top part of the device.
- Next Calibration Date (Validity): 02/2025.
- Calibration Date Performed: 02/2024.
This information indicates that the device was last calibrated in February 2024 and has a valid calibration certificate until February 2025 (considering the date the visual was taken, the device’s calibration is valid or nearing expiry).
Potential Fault Risk and Evaluation
Although the product is in good cosmetic condition and its calibration label is current (or recent), the “HW Error” warning appearing on the screen must be taken seriously. This error code may indicate faults in the flow sensor, turbine, or internal electronic board. Before being put into clinical use, the device must undergo “Error Code Analysis” and be repaired by an authorized technical service. Safe operation of the device cannot be guaranteed without resolving the hardware error.