Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 03.06.2026
Report code: 1780469871
FISHER & PAYKEL MR850 Heated Humidifier Analysis Report
Device Identification and Components
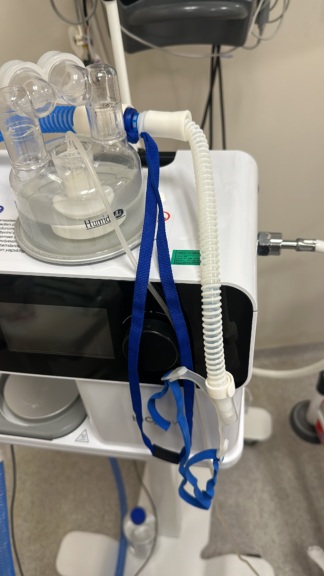
The images show a heated humidifier device used in respiratory support systems, mounted on a mobile stand, along with its accessories. The device consists of the main unit, a water chamber, a breathing circuit hose, a power cable, and a mobile wheeled stand.
Brand and Model Information
The FISHER & PAYKEL brand is clearly legible on the main unit of the device and on the base of the mobile stand. The MR850 model code is also clearly indicated on the main unit.
Authenticity Assessment
Upon examining the overall build quality, the clarity of logos and labels, the appearance of plastic components, and assembly details, the impression is that the device is an original production.
Usage Areas
The device in the image is a medical humidifier designed to heat and humidify gases administered to patients through the respiratory tract. Such devices are used for patients requiring mechanical ventilation, positive pressure respiratory support, and other medical gas applications. The fact that the device is on a mobile stand indicates portability and flexibility for different clinical environments or home use.
Quantity Information
- Main Heated Humidifier Unit: 1 piece
- Mobile Wheeled Stand: 1 piece
- Water Chamber: 1 piece (attached to the device)
- Breathing Circuit Hose: 1 piece
- Power Cable and Connection Cables: Several pieces
General Status and Condition
The general condition of the device is good, giving the impression of a used product. No significant wear or heavy damage has been observed. It is considered to be clean and well-maintained.
Physical Deformations
There are no visible major physical deformations such as large scratches, dents, cracks, or discoloration on the casing, mobile stand, or other visible surfaces of the product. Slight dust may be visible on the surfaces.
Mechanical Component Inspection
The wheels of the mobile stand appear sturdy. The connection points of the device to the stand and the height adjustment mechanism appear visually sound. No fractures or cracks have been detected in the plastic components.
Electronic Component Status
The power and sensor cables connected to the main unit of the device are positioned neatly. No obvious electronic component issues such as breaks, crushing, or fraying have been observed in the cables.
Condition of Accessories
The mobile stand, one breathing circuit hose, and power cables are provided with the device. The water chamber is attached to the device. There is no evidence in the images suggesting that any accessories are missing. No expiration date information is visible on the accessories. There are various socket inputs on the device; these inputs appear clean and undamaged.
Battery Status
No internal or external battery belonging to the device has been detected in the images. It is understood that the device is powered by the electrical grid.
Label Information
The following information and symbols are visible on the main unit of the device:
- FISHER & PAYKEL Healthcare Logo: Represents the manufacturer’s brand.
- MR850: The model code of the device.
- Heated Humidifier: Indicates that the device is a heated humidifier.
- Power Symbol: Indicates that the device operates on electricity.
- Warning Symbols: Indicate that the user manual should be consulted or that special attention is required.
Detailed label information such as serial number, lot number, or REF code is not clearly legible in the images.
Screen Analysis
There is a screen area on the main unit of the device. However, it is observed in the images that this screen is off or does not display any information, warnings, or values.
Size and Compatibility
The device is positioned on a mobile stand, which indicates that it can be easily moved and is suitable for use in different patient rooms or treatment areas. Its dimensions suggest it is a compact medical device suitable for bedside or patient-side use.
Production Year
There is no direct label or document regarding the production year of the device in the images. The general design and physical condition of the device point to a modern but not necessarily new-generation production period. This means the device may have been produced within the last few years, but a precise year cannot be specified.
Documents
No invoices, warranty certificates, user manuals, or other documents belonging to the device are visible in the images.
Usage Duration
No information regarding usage hours or total operating time has been detected on the device’s screen, labels, or any documents in the images.
Existing Faults
There is no evidence in the images pointing to any obvious faults such as broken parts, error messages, severed cables, or missing components on the device.
Potential Fault Risk
The general condition of the device is good. No concrete situation that could pose a potential fault risk, such as severe wear, rusting, or cable crushing, has been observed in the images.