Ready For Sale
Secondhand Philips Respironics Trilogy Evo Home Mechanical Ventilator
Price: USD$ 4.000,00 Approx: 180.000,00 TL
Ready For Sale
Ask a Question
Payment
No additional fees, full assurance. We provide complete financial and operational security in secondhand medical device trading. For this, we offer the "Secure Payment" service. This free service protects the rights of both parties by securing the buyer's money and the seller's product. The Secure Payment system is a standard assurance mechanism offered by Medbidding. For additional information, review the "services" page.
There is no cash on delivery order system on the Medbidding platform. For payments to be made by credit card, the product to be purchased must comply with this payment method. You can contact us to get information about this. We would be happy to assist you.
For payments made outside of Turkiye, you can choose bank transfer, credit card, Western Union or cryptocurrency options. Installment options are not currently available for credit cards other than Turkish banks.
Shipping
Free Shipping Conditions
You can benefit from our FREE SHIPPING campaign for your purchases in this category. Medbidding covers all shipping costs for both the buyer and the seller on orders that meet the conditions below. For all international sales and domestic sales within Turkiye that do not meet these conditions, Medbidding’s standard shipping procedures will apply.
Conditions
- The product must be sold for $400 USD or more.
- Both the buyer and the seller must be located in Turkiye.
Standard Shipping Conditions
In order to ensure secure transactions on Medbidding, the shipping process is managed through four different scenarios depending on the location of the buyer and seller. Free shipping is available for some categories. The terms below apply to all categories unless otherwise stated.
Buyer and Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer Outside Turkiye, Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer in Turkiye, Seller Outside Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer and Seller Outside Turkiye
If there is a local operation center in the seller's country:
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
If there is no operation center in the seller's country:
- Technical Inspection: Inspections are performed by our engineers via remote video call.
- Seller → Buyer: The seller packages the product and sends it directly to the buyer's address. The seller is responsible for this shipping cost.
Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 11.03.2026
Report code: 1773237899
Philips Respironics Trilogy Evo Analysis Report
Device Identification and Brand Model Information
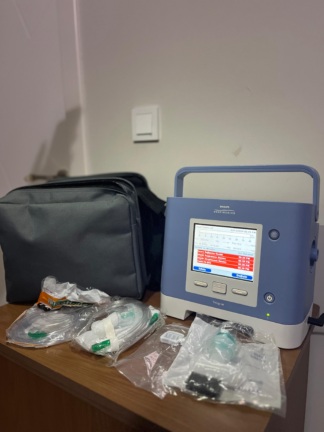
The device shown in the images is a respiratory support (mechanical ventilator) device. According to the inscriptions on the front body and screen interface of the device, its brand has been identified as Philips Respironics, and its model as Trilogy Evo. The model number 2110X15 is stated on the device’s system information screen.
Originality and General Condition
Upon examining the device’s casing structure, screen interface quality, brand logos, and menu layout, it is assessed to be an original product. The device is generally clean, well-maintained, and in high cosmetic condition.
Physical and Mechanical Assessment
No scratches, dents, cracks, dirt, or discoloration have been observed on the device’s white outer casing. The physical buttons on the front panel (alarm mute and power button) are in good condition. There is no visible deformation in the device’s mechanical or plastic components.
Electronic Components and Screen Analysis
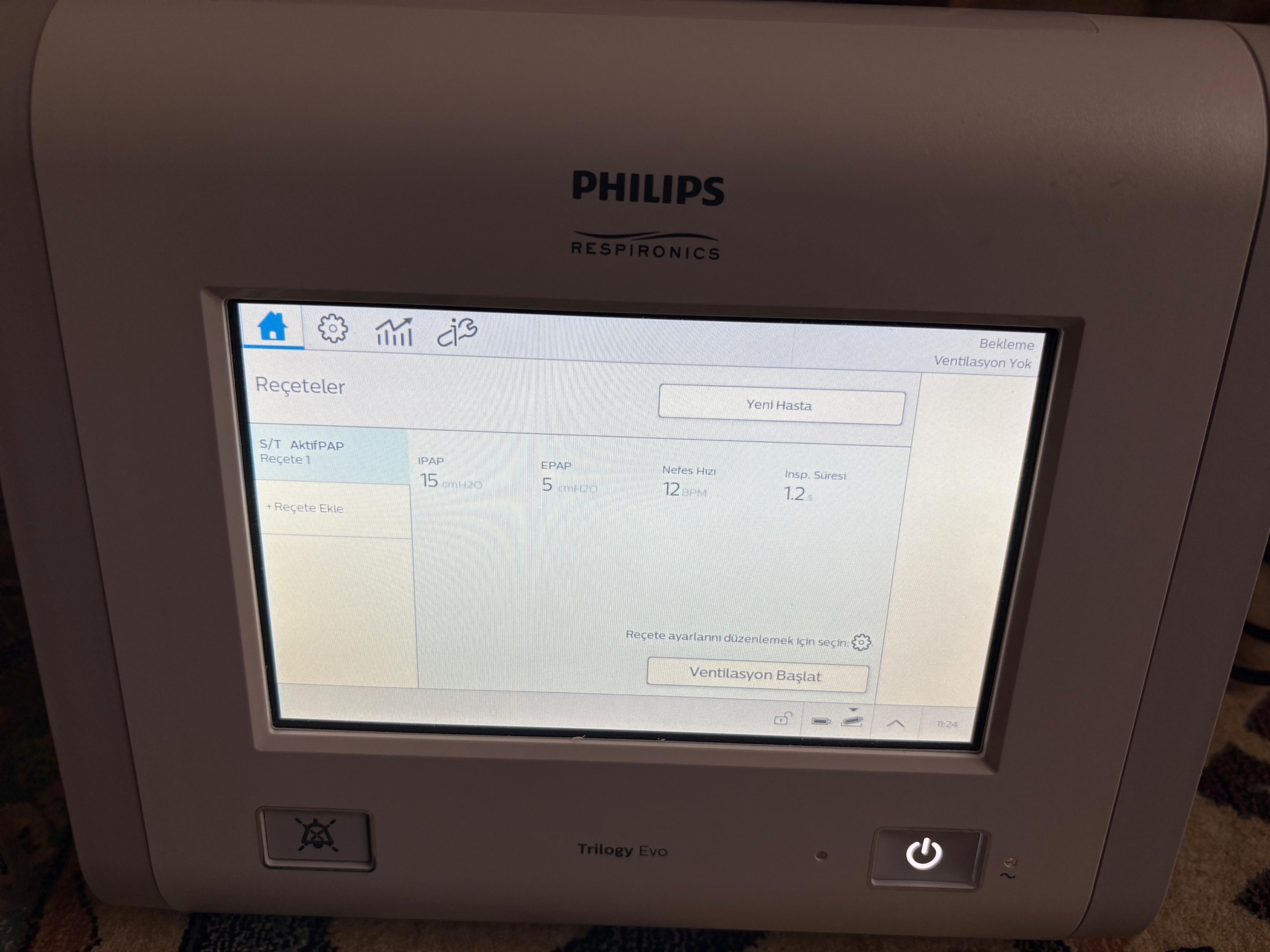
The device’s color digital touchscreen is active and functional. There is no pixel loss, scratches, or stains on the screen. The screen interface is in Turkish. The notifications and menu contents on the screen are as follows:
- Device status appears as Standby, No Ventilation.
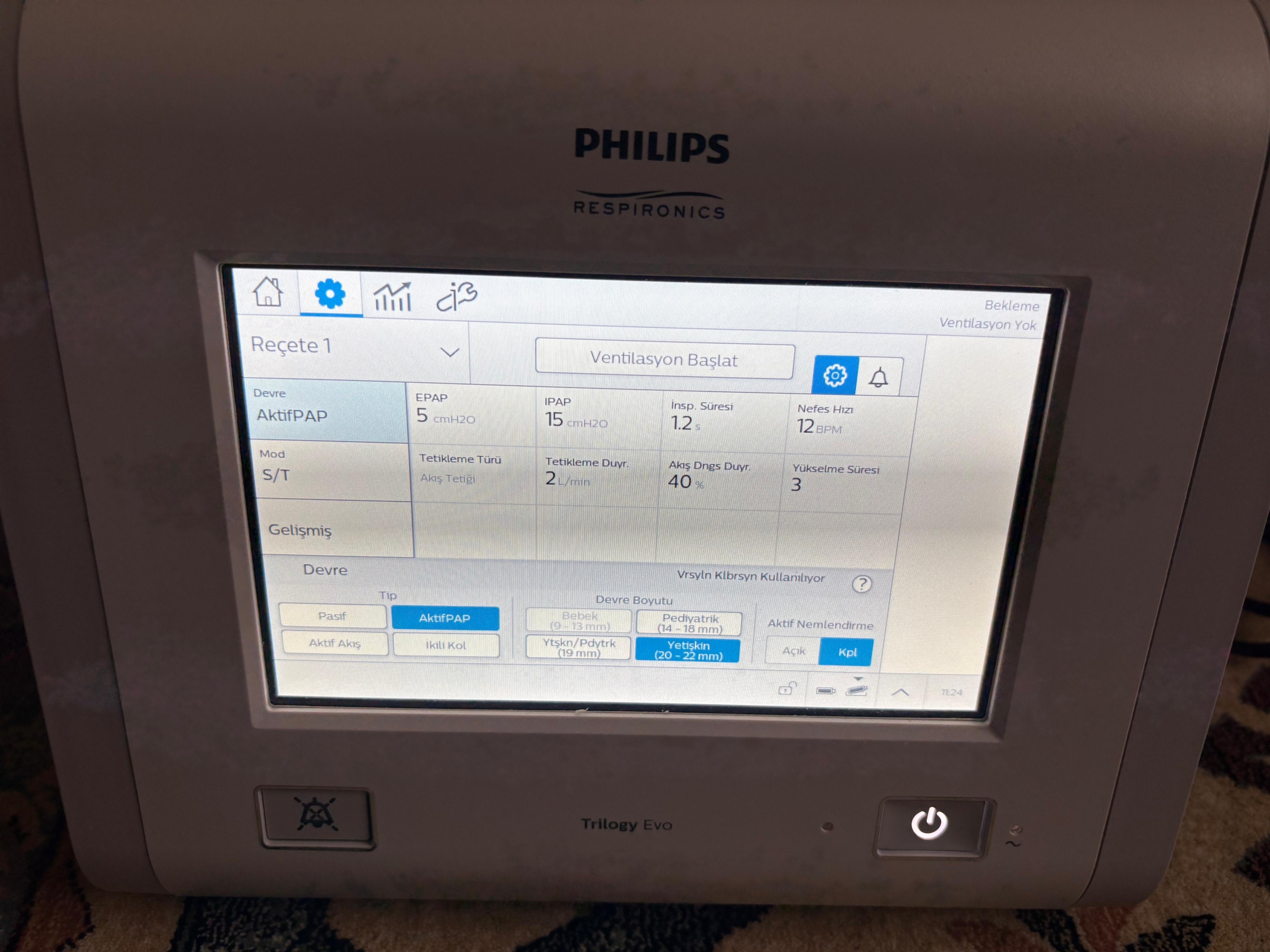
- In prescription settings, S/T mode and ActivePAP circuit are selected.
- Pressure settings are set to IPAP: 15 cmH2O and EPAP: 5 cmH2O.
- Breath rate is set to 12 BPM, inspiration time to 1.2 s.
- In advanced settings, trigger type is set to Flow Trigger, trigger sensitivity to 2 L/min, flow cycle sensitivity to 40%, and rise time to 3.
- On the Trends screen, the device’s system time appears as 02/10/2026 10:24:31 AM.
System and Label Information
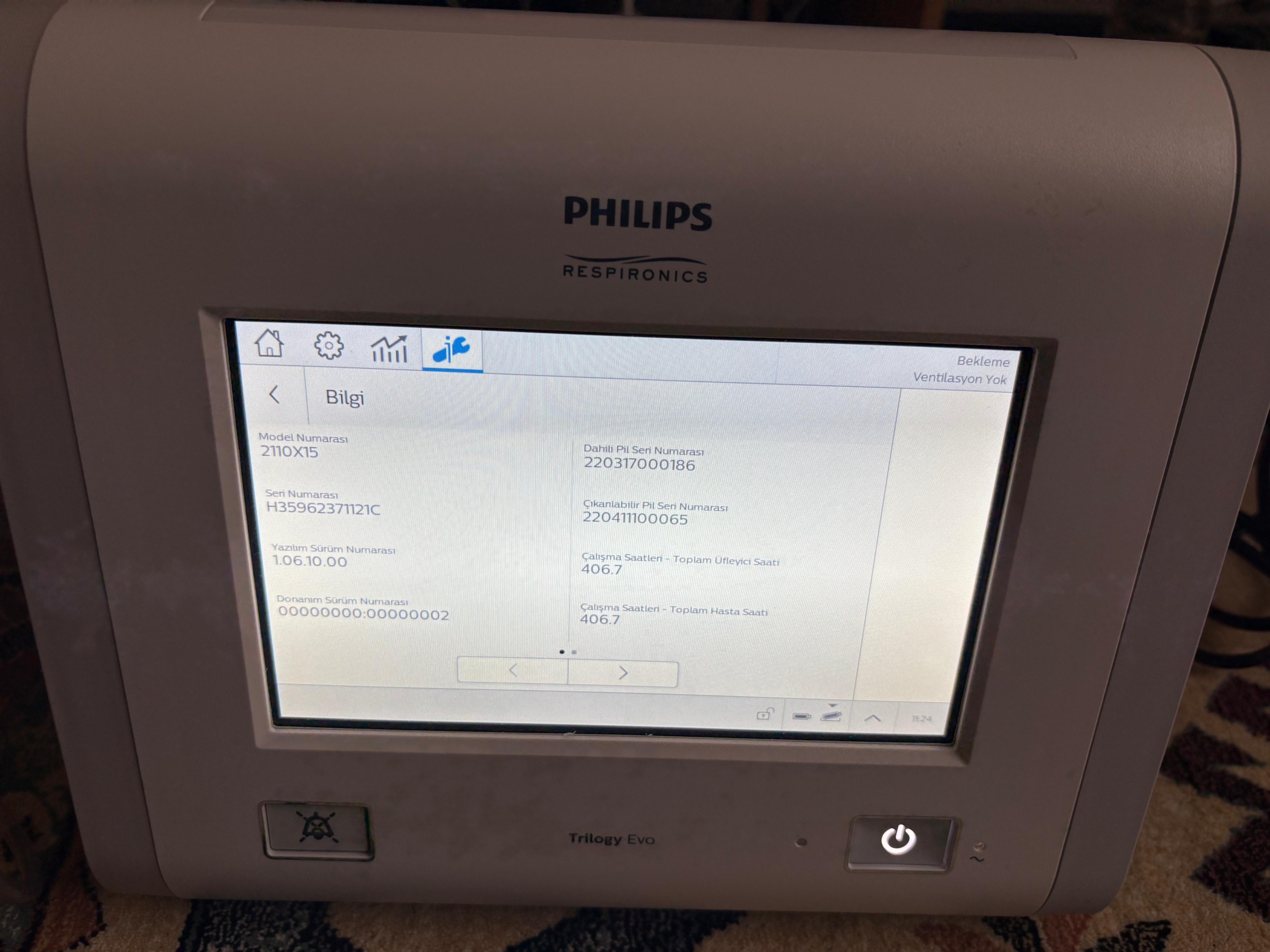
The system data obtained from the device’s information menu are listed below:
- Model Number: 2110X15
- Serial Number: H35962371121C
- Software Version Number: 1.06.10.00
- Hardware Version Number: 00000000:00000002
- Internal Battery Serial Number: 220317000186
- Removable Battery Serial Number: 22041100065
Usage Duration and Battery Status
According to system information, the device’s Total Blower Hours 406.7 and Total Patient Hours 406.7 have been recorded. These data indicate that the device has been used sparingly. It has been confirmed from the system menu that the device has both an internal and a removable battery. Battery icons are active in the status bar at the bottom of the screen. There is no externally observable battery swelling or physical damage.
Accessories and Quantity Information
A total of 3 main parts have been identified in the images:
- 1 main device (Philips Respironics Trilogy Evo)
- 1 gray and black original carrying case with a Philips logo
- 1 transparent, corrugated breathing circuit (hose). This hose has a green bacterial/viral filter and a white fabric fastening apparatus at its end.
No tearing, breakage, or deformation has been observed in the accessories.
Areas of Use and Compatibility
This device is used to provide mechanical ventilation to patients requiring respiratory support. In the device’s menu, the circuit size option Adult (20 – 22 mm) is selected. Additionally, Baby (9 – 13 mm) and Pediatric (14 – 18 mm) options are also available in the menu. This indicates that the device is compatible with patients of different age groups when appropriate breathing circuits are used.
Malfunction Status and Potential Risks
No error messages, broken parts, or evidence of an existing malfunction related to the device are found in the images. The device’s general condition, outer casing, and hose connections are in very good condition. Based on the observed physical condition, no potential malfunction risk has been identified in the device.