Ready For Sale
Secondhand Edan M8 Patient Monitor
Price: USD$ 500,00 Approx: 22.500,00 TL
Ready For Sale
Ask a Question
Payment
No additional fees, full assurance. We provide complete financial and operational security in secondhand medical device trading. For this, we offer the "Secure Payment" service. This free service protects the rights of both parties by securing the buyer's money and the seller's product. The Secure Payment system is a standard assurance mechanism offered by Medbidding. For additional information, review the "services" page.
There is no cash on delivery order system on the Medbidding platform. For payments to be made by credit card, the product to be purchased must comply with this payment method. You can contact us to get information about this. We would be happy to assist you.
For payments made outside of Turkiye, you can choose bank transfer, credit card, Western Union or cryptocurrency options. Installment options are not currently available for credit cards other than Turkish banks.
Shipping
Standard Shipping Conditions
In order to ensure secure transactions on Medbidding, the shipping process is managed through four different scenarios depending on the location of the buyer and seller. Free shipping is available for some categories. The terms below apply to all categories unless otherwise stated.
Buyer and Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer Outside Turkiye, Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer in Turkiye, Seller Outside Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer and Seller Outside Turkiye
If there is a local operation center in the seller's country:
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
If there is no operation center in the seller's country:
- Technical Inspection: Inspections are performed by our engineers via remote video call.
- Seller → Buyer: The seller packages the product and sends it directly to the buyer's address. The seller is responsible for this shipping cost.
Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 03.04.2026
Report code: 1775209835
EDAN M8 Patient Monitor Analysis Report
Device Identification and Areas of Use
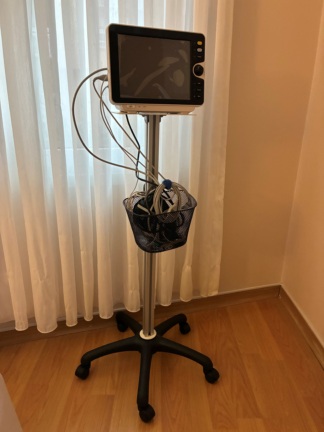
The product in the image is a patient monitor. These devices are suitable for use in hospital emergency rooms, intensive care units, operating theaters, and inpatient wards to instantly monitor patients’ vital signs.
Brand and Model Information
According to the written logo located in the upper left corner of the device’s front panel, its brand is EDAN, and its model extension is identified as M8. Upon examining the device’s casing structure, button layout, and logo print quality, the product is assessed to be original.
Quantity Information
There is 1 main device in the image. No power cable, measurement probe, or external accessories belonging to the device are visible in the image.
General Condition and Physical Deformations
The device’s outer casing is made of white hard plastic. No cracks, fractures, dents, deep scratches, or yellowing have been observed on the casing. The device’s overall condition is very clean and good. A dotted surface texture is present on the right and left edges of the screen, providing ease of grip.
Screen Analysis
A large digital screen is located on the front surface of the device. Since the device is turned off, no data or menu is visible on the screen. A transparent factory protective film, which has started to peel off from its upper right corner, is present on the screen. Slight dust and stain marks are visible on the outer surface of the film, but no physical damage or crack has been detected on the main screen panel underneath.
Mechanical and Electronic Components
Control buttons and indicators are located on the lower part of the device’s front panel. There is no fading, depression, or physical deformation on the buttons. The components located on the front panel are as follows:
- One power (on/off) button on the far left.
- CHARGE and ALARM notification lights (LEDs) immediately to the right of the power button.
- Six membrane-type function buttons in the middle section, featuring various medical symbols (ECG waveform, blood pressure cuff, alarm mute, print, etc.).
- One blue rotary control knob in the lower right corner, used for menu navigation and confirmation.
Since the rear, bottom, or side panels of the device are not visible in the image, there is no data regarding battery status, label information (serial number, manufacturing year, etc.), and sensor socket inputs.
Potential Malfunction Risk
During the examination conducted via the image, no impact, liquid contact traces, or structural defects that could cause a malfunction have been observed on the device’s outer surface, screen, or keypad. The device’s physical condition maintains its integrity.