Ready For Sale
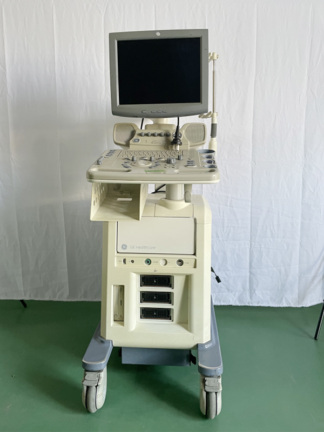
Secondhand Aloka Flexus SSD-1100 Ultrasound Machine
Price: USD$ 2.200,00 Approx: 99.000,00 TL
Ready For Sale
Ask a Question
Payment
No additional fees, full assurance. We provide complete financial and operational security in secondhand medical device trading. For this, we offer the "Secure Payment" service. This free service protects the rights of both parties by securing the buyer's money and the seller's product. The Secure Payment system is a standard assurance mechanism offered by Medbidding. For additional information, review the "services" page.
There is no cash on delivery order system on the Medbidding platform. For payments to be made by credit card, the product to be purchased must comply with this payment method. You can contact us to get information about this. We would be happy to assist you.
For payments made outside of Turkiye, you can choose bank transfer, credit card, Western Union or cryptocurrency options. Installment options are not currently available for credit cards other than Turkish banks.
Shipping
Standard Shipping Conditions
In order to ensure secure transactions on Medbidding, the shipping process is managed through four different scenarios depending on the location of the buyer and seller. Free shipping is available for some categories. The terms below apply to all categories unless otherwise stated.
Buyer and Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer Outside Turkiye, Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer in Turkiye, Seller Outside Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer and Seller Outside Turkiye
If there is a local operation center in the seller's country:
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
If there is no operation center in the seller's country:
- Technical Inspection: Inspections are performed by our engineers via remote video call.
- Seller → Buyer: The seller packages the product and sends it directly to the buyer's address. The seller is responsible for this shipping cost.
Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 27.11.2025
ALOKA Medical Ultrasound Imaging System Analysis Report
Device Detection and Identification
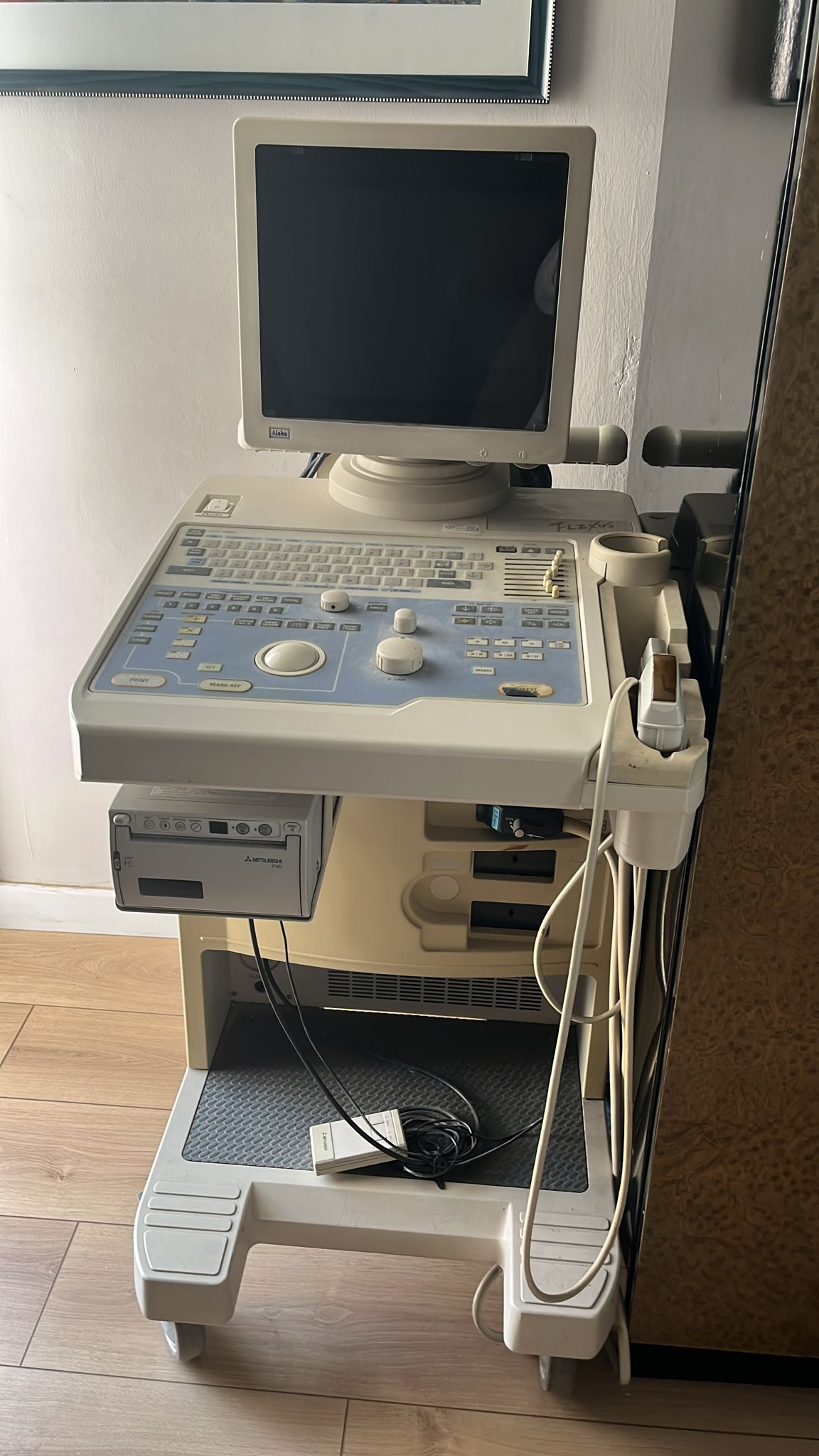
The device shown in the visuals is a mobile ultrasonography (ultrasound) device used for medical diagnostic purposes. The unit consists of an integrated CRT monitor, a comprehensive control panel, probe (transducer) holders, an external printer for data/image output, and a wheeled stand structure for transport. The system is designed as an integrated station.
Brand and Model Information
Upon examining the visual details on the product, the following brand identifications were made:
- Device Brand: A clear ALOKA logo is visible in the lower-left corner of the monitor frame on the top section. The inscription “FLEXUS” is seen handwritten on the upper right body of the device; while this inscription might suggest that the device belongs to the “Aloka SSD-1100 Flexus” series, it is noted as visual data rather than definitive model information, as it is not a factory-printed label.
- Printer Brand: The MITSUBISHI logo is clearly readable on the thermal video printer located on the bottom shelf of the device.
Areas of Use
This device is used in medical specialties such as radiology, obstetrics, gynecology, urology, and internal medicine for imaging internal organs, soft tissues, and blood flow (if Doppler feature is present). The mobile nature of the device allows it to be transported to different rooms within the hospital or to the bedside.
Quantity Information
The contents of the set seen in the visual are as follows:
- 1 Main Ultrasound Unit (Including monitor and console)
- 1 Integrated Thermal Printer (Mitsubishi brand)
- 1 Ultrasound Probe (Attached to the right side of the device)
- 1 Footswitch/Remote Control (Placed on the bottom shelf)
General Condition and Status Assessment
The device is in a used condition and shows clear signs of aging. The product is not new and features technology old enough to be classified as “vintage” (CRT monitor, floppy disk drive, etc.). According to visual inspection, the physical condition of the device is moderate.
Physical Deformation and Cosmetic Condition
Notable physical conditions on the device include:
- Discoloration (Yellowing): There is a significant level of yellowing on the device’s lower plastic body, particularly on the foot sections, due to exposure to time and UV light. Although the monitor casing remains whiter than the body, tonal differences have occurred.
- Writing and Markings: “FLEXUS” is written with a ballpoint or marker pen on the upper right corner of the main console. This is not an original labeling.
- Dirt and Dust: Accumulated dust and dirt are present between keyboard keys, around the wheels, and on cable bundles.
Electronic and Mechanical Component Analysis
- Monitor: An old-style (CRT) screen is located on the upper part of the device. Since the screen is off, no comment can be made regarding pixel errors or its operational status.
- Control Panel: The QWERTY keyboard, TGC (Time Gain Compensation) slider adjustment knobs, trackball, and various function keys are in place. No excessive wear is visible on the keys, but there are signs of use around the trackball.
- Mechanical Structure: The wheeled transport stand appears sturdy. The printer bay and cable arrangements on the device’s bottom shelf are present.
Accessories and Connections
Accessory details identified in the visual:
- Ultrasound Probe: 1 probe is visible, placed in the holder on the right side of the device. The probe’s cable hangs downwards, and no visual breakage has been detected in the cable.
- Thermal Printer: A Mitsubishi brand video printer (copy processor) is mounted on the lower-left shelf of the device. The buttons on the front panel appear intact.
- Footswitch: A wired remote control/footswitch, likely belonging to the Mitsubishi printer, is located on the bottom carrier tray.
- Cables: Power and probe cables hanging from the back and sides of the device are disorganized. No significant crushing or breakage in the cables can be visually distinguished, but they have a tangled appearance.
Technical Details and Observations
- Floppy Drive: A 3.5-inch floppy disk drive or a similar recording unit bay is visible directly above the printer, which confirms the device’s older manufacturing year.
- Panel Details: Standard ultrasound control terms such as “Active”, “Freeze”, and “Mark Ref” are legible on the control panel.
Originality Status
The Aloka logo on the product, the integrated Mitsubishi printer, and the typical medical device design indicate that the product is an original Aloka ultrasound device. No subsequently added or modified parts (except for the handwriting) have been observed.
Potential Malfunction Risk and Warnings
Given the age of the device and the technology used (CRT screen, old-style data storage), there is a high probability that electronic components (capacitors, etc.) have reached the end of their lifespan. Furthermore, while the risk of oxidation due to long-term storage for the probe cable and connection points cannot be visually confirmed, it should be taken into consideration. No physically broken parts are visible, but a guarantee of “working condition” cannot be provided based on the visuals.