Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 01.12.2025
Mindray DP-6600 Digital Ultrasonic Imaging System Analysis Report
Device Detection and Identification
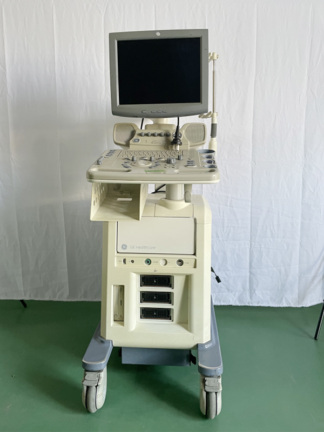
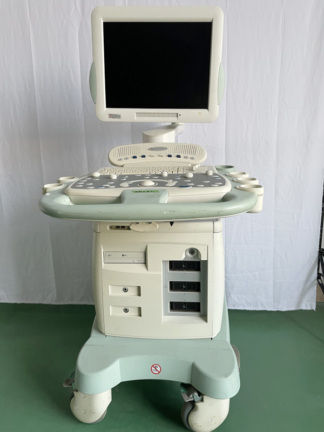
The product shown in the visuals is a portable ultrasound device used for medical diagnosis and imaging. The device consists of a main body, a foldable monitor unit, a control panel (keyboard), and an integrated ultrasound probe. The gridded structure on the device’s back panel is part of the ventilation system. This is not a patient bed or a transport lift; it is directly identified as a “Digital Ultrasonic Diagnostic Imaging System”.
Brand and Model
As a result of detailed examinations, the device’s brand has been definitively identified as Mindray and its model as DP-6600. The inscription Digi Prince DP-6600 is clearly visible on the lower left part of the screen frame, immediately below which the manufacturer’s logo and brand MINDRAY are clearly readable. Furthermore, the label on the back of the device clearly states “Model: DP-6600”. This identification is 100% conclusive.
Label Information and Technical Data
The following information has been read in detail and transcribed from the technical label on the back of the device:
- Device Description: Digital Ultrasonic Diagnostic Imaging System
- Model: DP-6600
- Serial Number (SN): BE55-0265
- Date of Manufacture: 2005-05 (May 2005)
- Lot/Version Information: S06, ver1.0
- Power Ratings: 100-240V~ 50/60Hz 150VA
- Other Codes: The label also contains the inscription “BE55-0265” directly below the barcode, the “CE 0123” certification mark, and European Representative (EC REP) information.
Manufacturer Information
“Shenzhen Mindray Bio-Medical Electronics Co.,Ltd.” appears as the manufacturer on the label. The place of manufacture is stated as China (Shenzhen). The European representative is recorded as “Shanghai International Holding Corp. GmbH(Europe)” with an address in Hamburg, Germany.
General Condition and Physical State
The device is considered a used product based on visual inspection. Its outer casing is beige/cream colored, and slight age-related discoloration (yellowing) can be observed in the plastic components. However, no deep cracks, breaks, or major physical damage have been detected on the casing. The foldable mechanism appears robust, and the handle and probe holder attachments are in place. The device generally maintains its integrity.
Electronic and Control Panel Status
The device’s control panel appears complete. QWERTY keyboard keys, TGC (Time Gain Compensation) sliders (6 units), trackball (cursor ball), Gain adjustment knob, and other function keys (Probe, Freq, Menu, Print, Freeze, etc.) are physically present. The inscriptions on the keys are legible; no excessive fading is observed. However, as the device is powered off, whether the screen is working, if there are pixel errors, or if the system boots up cannot be determined visually.
Mechanical Components
The hinges that allow the screen to open and close appear visually sound. The carrying handle on the top is not deformed. No broken slats are visible in the rear ventilation grilles; the grilles are intact.
Accessories and Connections
1 ultrasound probe (transducer) connected to the device is visible in the visual. The probe’s cable is plugged into the device and placed in the holder on the right side. Although there are slight bends in the cable, no clear tear is observed in its outer insulation. Apart from the probe, no additional accessories such as a power cable, user manual, or gel are present in the visual.
Areas of Use
This product is used in medical institutions (hospitals, clinics) for imaging internal body structures (organ, tissues) with ultrasound waves. It is a fundamental imaging device that aids in diagnosis in specialties such as abdominal, obstetrics, and urology. The Mindray DP-6600 model, thanks to its portable design, is suitable for use in mobile healthcare services or bedside applications.
Originality
Upon examining the quality of the labels, logo printing, keypad layout, and material structure on the device, it is understood that the product is an Original Mindray production. There is no indication of it being a counterfeit or replica.
Year of Manufacture and Age
According to the clear information on the label, the product was manufactured in May 2005. As of the report date, it is approximately 20 years old. This indicates that it is an older generation (Digital Ultrasonic) device compared to technologically current devices.
Quantity Information
The visuals show 1 main unit of Mindray DP-6600 and 1 connected probe.
Potential Failure Risk and Assessment
As a result of the visual analysis, no “broken/fragmented” area that would prevent use is observed on the device’s casing or buttons. However, due to the product being manufactured in 2005, there is a risk that its electronic components (especially motherboard capacitors, CRT/LCD screen lifespan) may be nearing the end of their natural life. The trackball mechanism may lose its sensitivity over time due to dust accumulation. Mechanically and cosmetically, it is in “Good/Acceptable” condition for its age.