Ready For Sale
Secondhand GE Voluson E6 Ultrasound Machine
Price: USD$ 25.000,00 Approx: 1.125.000,00 TL
Ready For Sale
Ask a Question
Payment
No additional fees, full assurance. We provide complete financial and operational security in secondhand medical device trading. For this, we offer the "Secure Payment" service. This free service protects the rights of both parties by securing the buyer's money and the seller's product. The Secure Payment system is a standard assurance mechanism offered by Medbidding. For additional information, review the "services" page.
There is no cash on delivery order system on the Medbidding platform. For payments to be made by credit card, the product to be purchased must comply with this payment method. You can contact us to get information about this. We would be happy to assist you.
For payments made outside of Turkiye, you can choose bank transfer, credit card, Western Union or cryptocurrency options. Installment options are not currently available for credit cards other than Turkish banks.
Shipping
Standard Shipping Conditions
In order to ensure secure transactions on Medbidding, the shipping process is managed through four different scenarios depending on the location of the buyer and seller. Free shipping is available for some categories. The terms below apply to all categories unless otherwise stated.
Buyer and Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer Outside Turkiye, Seller in Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer in Turkiye, Seller Outside Turkiye
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
Buyer and Seller Outside Turkiye
If there is a local operation center in the seller's country:
- Seller → Medbidding: The seller packages the product and sends it to the Medbidding operation center. The seller is responsible for this shipping cost.
- Medbidding → Buyer: The product, inspected by Medbidding engineers, is sent to the buyer's address. The buyer is responsible for this shipping cost.
If there is no operation center in the seller's country:
- Technical Inspection: Inspections are performed by our engineers via remote video call.
- Seller → Buyer: The seller packages the product and sends it directly to the buyer's address. The seller is responsible for this shipping cost.
Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 31.10.2025
GE Voluson E6 Ultrasound Device Analysis Report
Overview and Device Description
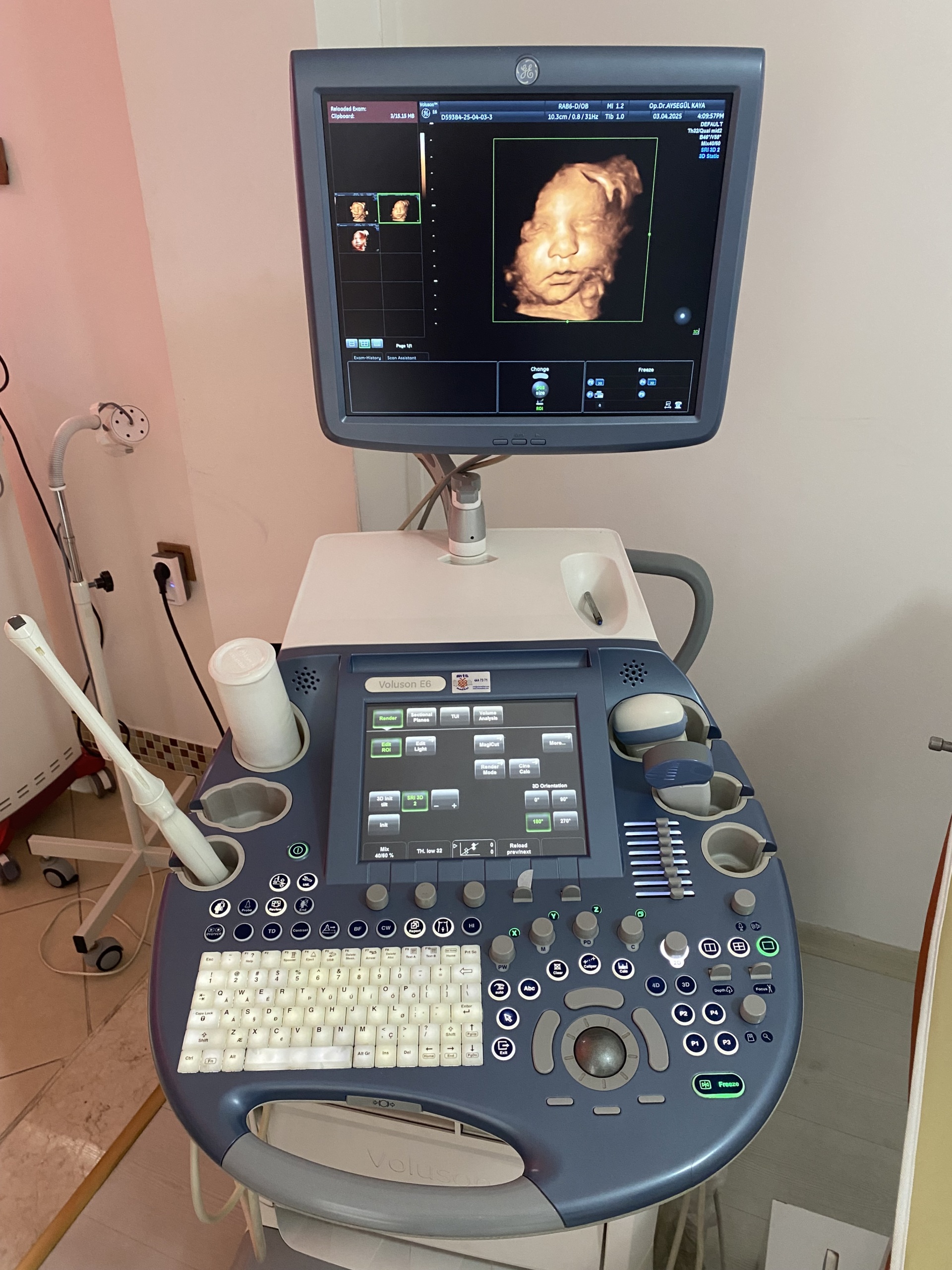
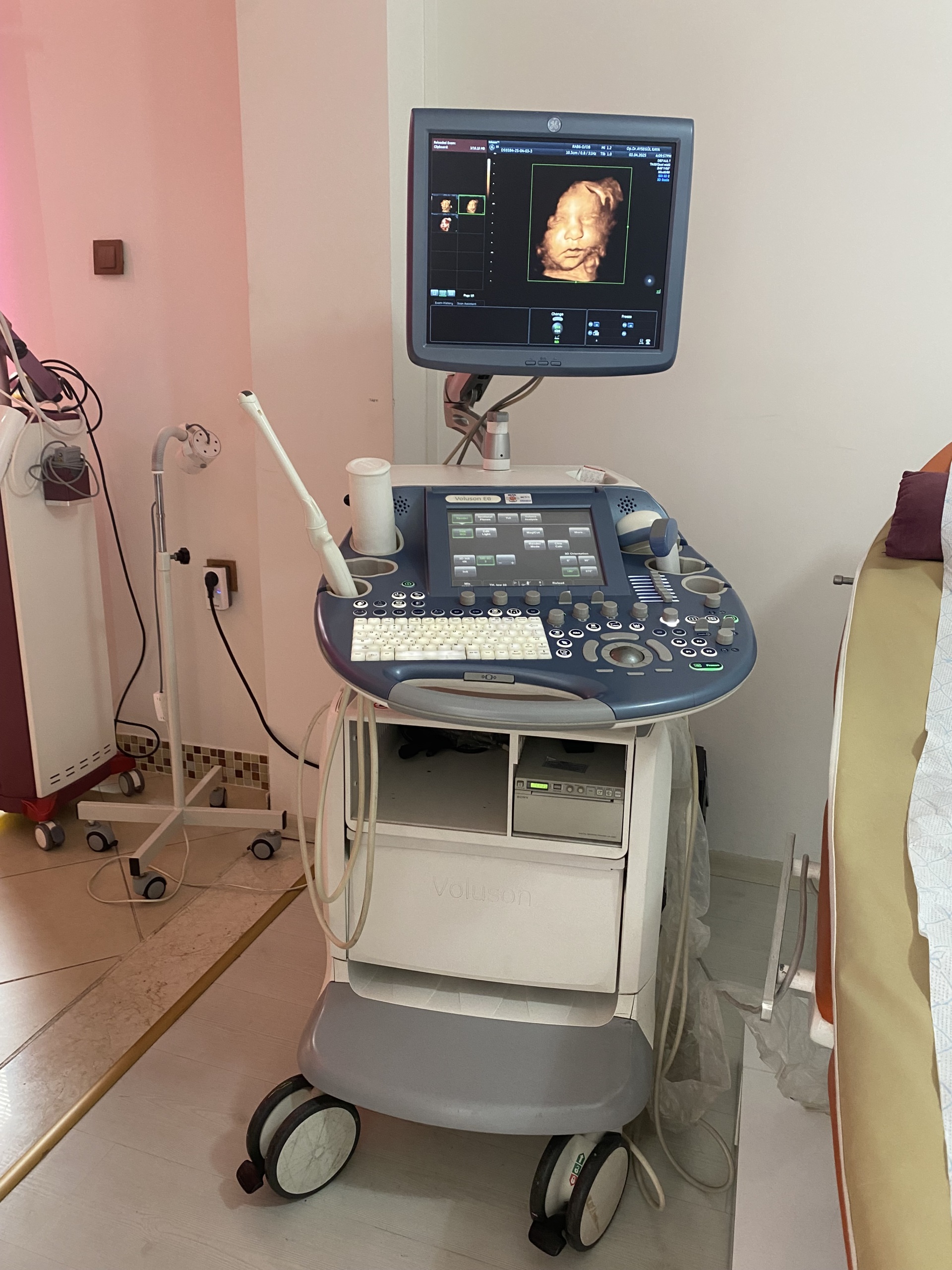
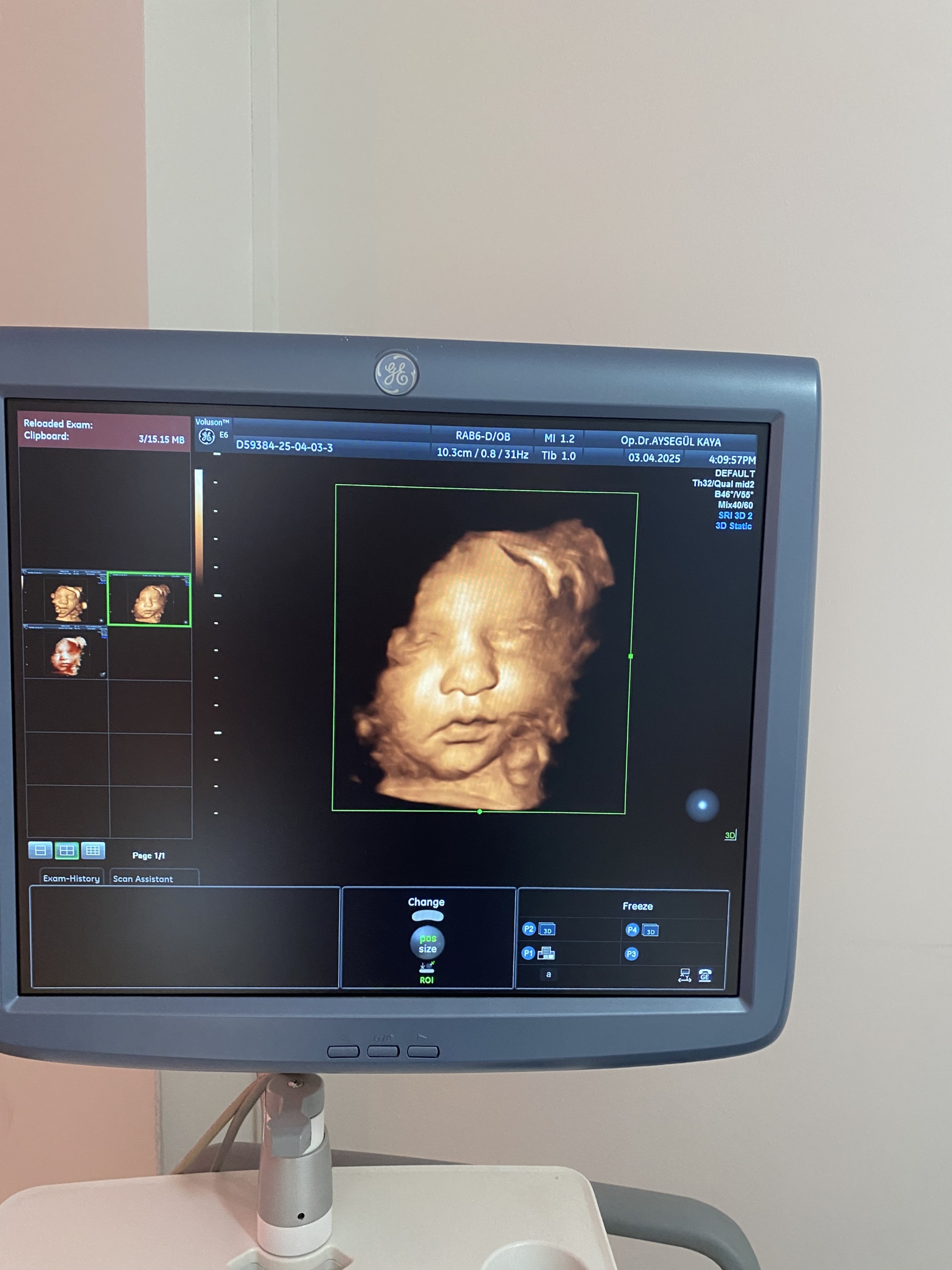
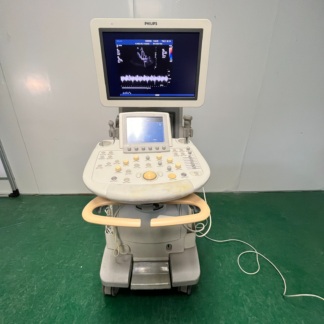
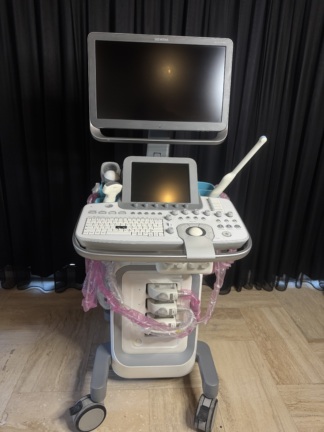
The product shown in the images is an ultrasound device used for medical diagnostic purposes, integrated onto a wheeled stand. The device consists of a main imaging monitor, a touch control panel, an alphanumeric keyboard, probe (transducer) slots, and at least one ultrasound probe. The 3D fetal (baby in the womb) images and the probe model visible on the screens indicate that the device is commonly used, especially in obstetrics and gynecology. However, it can also be used for general radiology and other medical imaging procedures by utilizing different probes.
The total number of devices in the images is 1. In addition to the device, at least two ultrasound probes and one ultrasound gel bottle are visible as accessories.
Brand and Model Information
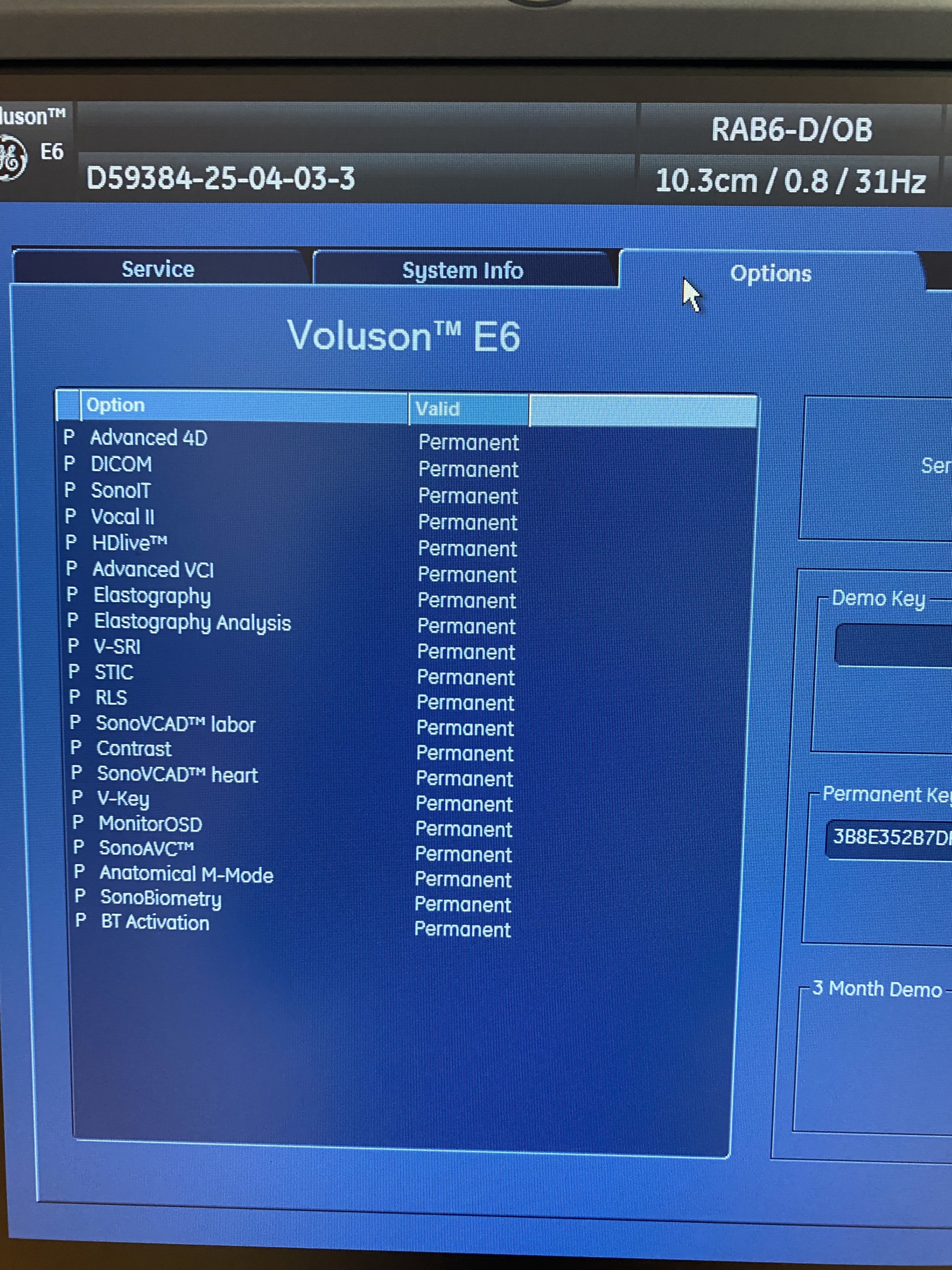
As a result of detailed visual inspection, the brand and model of the device have been clearly identified. A prominent GE (General Electric) logo is located on the frame of the main monitor and within the system software. The model name Voluson E6 is clearly written on the device’s control panel and casing. Furthermore, the system information screen also confirms the model as Voluson™ E6.
- Device Brand-Model: GE Voluson E6
- Probe Brand-Model: According to the screen data in the image, the model of the probe connected to the device is identified as RAB6-D/OB.
Physical Condition and Cosmetic Evaluation
Although the general condition of the device is used, it is observed to be in very good condition. Cosmetically, it has a clean and well-maintained appearance. There are no significant deformations such as serious scratches, dents, cracks, or discoloration on the casing, control panel, or monitor housing. Minimal signs of use may be visible due to mobile usage stemming from its wheeled structure, but its physical integrity is generally complete.
No visible breakage, missing parts, or functional issues were detected in the mechanical components such as the wheels, probe holders, the articulated arm supporting the monitor, and the keyboard mechanism. All buttons and control knobs are in place and do not show excessive wear.
Electronic Components and Functionality
The images show the device running and actively in use. Both the main monitor and the touch control panel screens are on and display images without issues. No pixel errors, darkening, or abnormalities were observed on the screen. A unit resembling a CD/DVD drive is located at the bottom of the device, and its cover is intact. This indicates that the device’s basic electronic components are functional.
Screen Analysis and Software Information
Important technical and software information is present on the device’s screens. A detailed analysis is as follows:
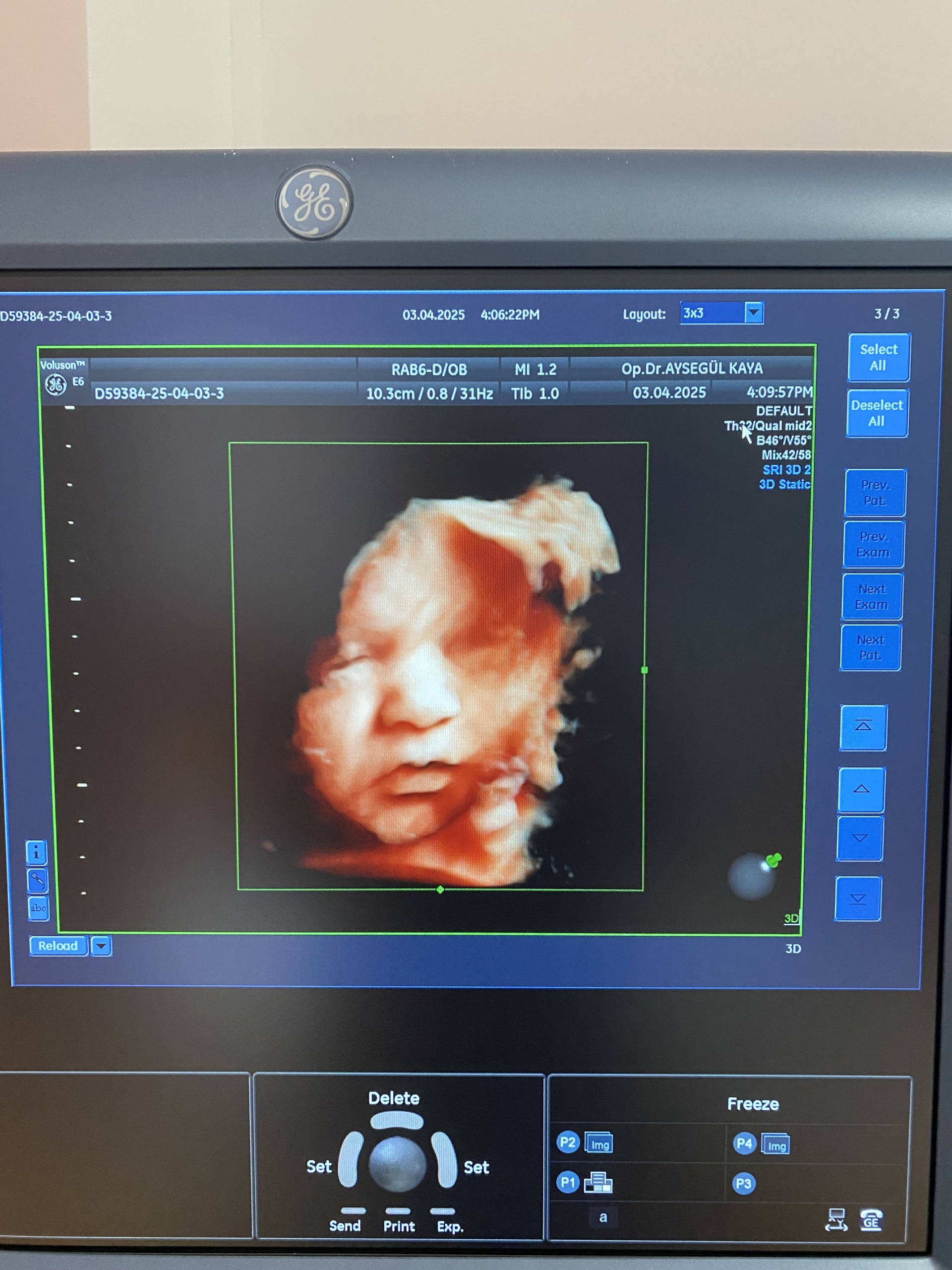
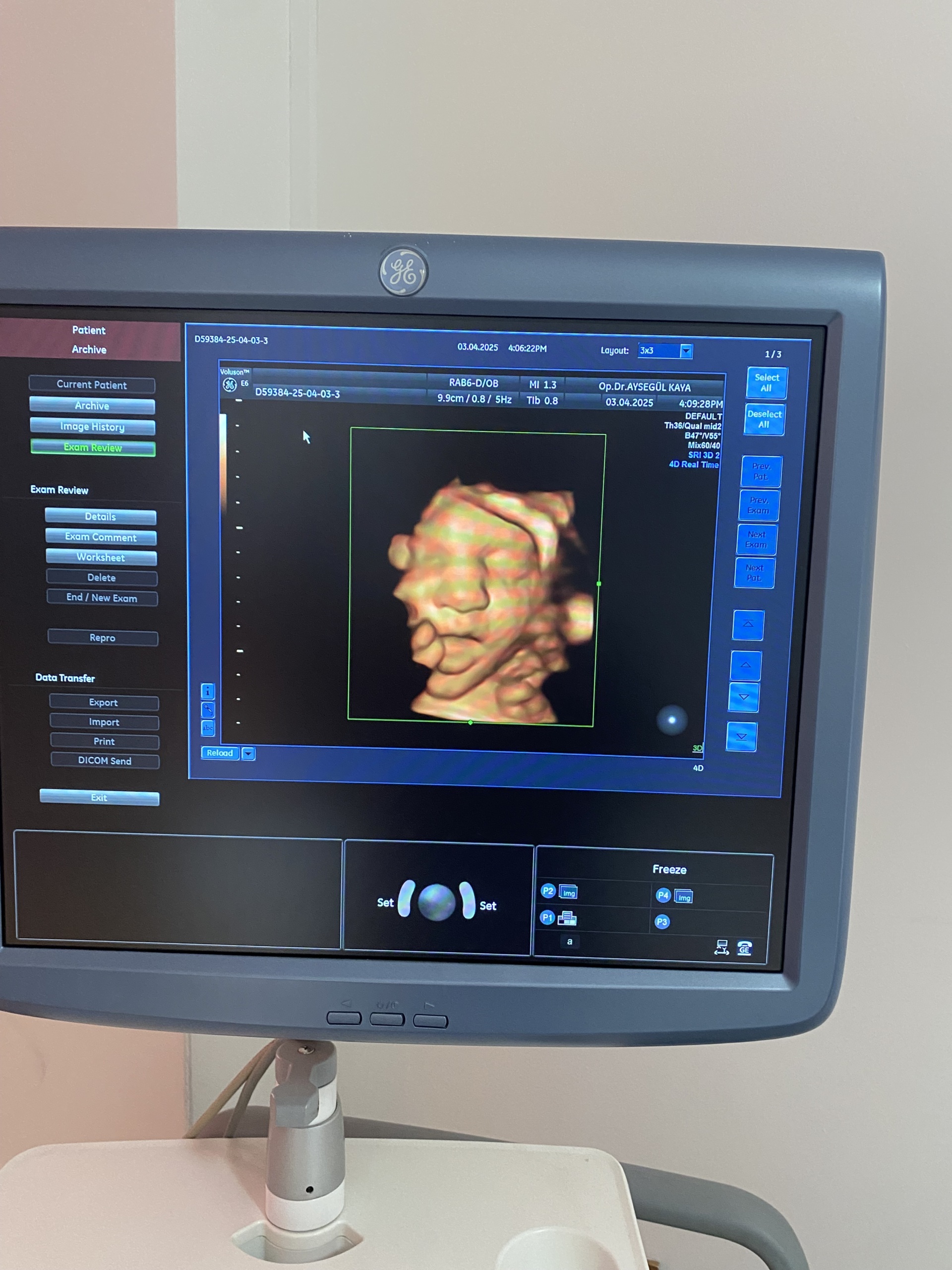
- Imaging: The screen successfully displays a 3D fetal ultrasound image. This proves that the device’s advanced imaging modes are active.
- Date Information: The system date 03.04.2025 is visible on the screen. Since this date indicates a future date compared to the report creation date, it is understood that the device’s system clock is not set correctly.
- Software Licenses: The “System Info” screen capture lists numerous software options permanently licensed on the device. These include important and valuable features such as Advanced 4D, DICOM, HDlive™, SonoIT, Elastography, V-SRI, and Anatomical M-Mode. The permanent nature of these licenses means that the features can be used without additional fees or time restrictions.
- Identifier Code: The code D59384-25-04-03-3 seen on the screens could be a patient or examination number, or it could be a system identifier. It is not labeled as a serial number.
Accessories
The accessories visible with the device are listed below:
- Ultrasound Probes (Transducers): At least two probes are present on the device. The model of one of these is stated as RAB6-D/OB in the screen data. No visible damage or wear is present on the cables or heads of the probes.
- Gel Bottle: One ultrasound gel bottle is present in one of the probe holders.
- Keyboard: An integrated, pull-out QWERTY keyboard is included with the device.
Potential Risk Assessment
Based on the available visuals, there is no clear evidence of any malfunction in the device. The device is in working condition and is physically well-preserved. No crushing of cables, cracks in the casing, or visible damage to the electronic components were detected. Therefore, based solely on visual evidence, the potential risk of malfunction is assessed as low. The device’s overall condition gives the impression that it has been regularly maintained and carefully used.