Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 13.05.2026
Report code: 1778677802
Toshiba Aplio 500 Ultrasound Device Analysis Report
Device Identification
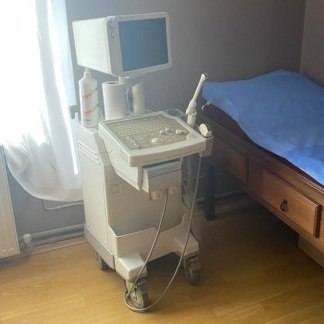
The product in the image is a mobile ultrasound device used for medical imaging purposes.
Brand and Model
The Toshiba brand is clearly legible on the upper part of the screen frame and in the top-left corner of the screen. On the main screen, the model name is visible in capital letters as Aplio 500. Based on this information, the product brand is Toshiba and the model is Aplio 500.
Authenticity
The quality of the logos on the device, the layout of the buttons on the control panel, the structure of the screen, and the general craftsmanship details indicate that the product is an original Toshiba Aplio 500 ultrasound device.
Usage Areas
This device is used in various departments of hospitals and clinics, such as radiology, cardiology, and obstetrics and gynecology, to image patients’ internal organs, tissues, and blood flow using a non-invasive method. Buttons on the control panel such as PW, CDI, ADF, and 2D indicate that different imaging modes are available.
Quantity Information
There is one complete ultrasound device in the image. No probes (transducers) or external accessories connected to the device are visible.
General Condition
The main screen of the device is on and operational. The control panel and touch screen maintain their integrity. The general condition of the device appears to be good.
Physical Deformation
No serious physical damage such as breaks, cracks, or deep dents has been observed on the device casing, screen frame, or control panel. The labels on the buttons of the control panel are legible. There is slight dust accumulation on the surfaces of the device due to usage.
Mechanical Components
The hinge structure of the main screen appears to be sturdy. The probe holders and gel bottle slots are in place. The wheels of the device cannot be fully distinguished in the image.
Electronic Components
The main screen of the device is active and displaying the startup screen. The surface of the touch screen on the upper left part of the control panel is intact. The keypad and trackball are in place as a whole.
Accessories
The device features probe holders and gel bottle slots. However, no ultrasound probes, power cables, or other accessories connected to the device are present in the images. The socket inputs on the sides of the device are not visible in the image.
Battery Status
There is a battery icon and light on the left side of the control panel. This indicates that the device may have an internal battery. However, no information regarding the capacity or physical condition (swelling, leakage, etc.) of the battery can be obtained from the image.
Label Information
There are no technical labels containing a serial number, lot number, or REF code on the front surface of the device.
Screen Analysis
The main screen of the device is on. The screen displays the Toshiba logo, the Aplio 500 model name, and a snowflake-like background image. Copyright information is located in the bottom-right corner of the screen. A gray rectangular area is visible in the center of the screen. The screen display indicates that the device software is loaded or it is in standby mode.
Dimensions and Compatibility
There is no information in the image regarding the physical dimensions of the product or which patient group (infant, child, adult) it is suitable for.
Year of Manufacture
The year of manufacture of the device cannot be determined from the labels or screen information in the image.
Documents
There are no documents such as invoices, warranty certificates, or user manuals belonging to the device in the images.
Usage Duration
There is no information on the screen or the body regarding the total operating hours of the device or how long it has been used.
Existing Faults
There is no clear evidence in the images, such as broken parts or error messages on the screen, indicating that the device is faulty. The fact that the screen is on indicates that the basic electronic components are working.
Potential Fault Risk
No negative conditions such as serious wear, rust, or damage to the cables have been observed on the device in the images. The physical condition of the device is good.