Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 30.05.2026
Report code: 1780100787
TOSHIBA Xario SSA-660A Ultrasound System Analysis Report
Device and Accessory Identification
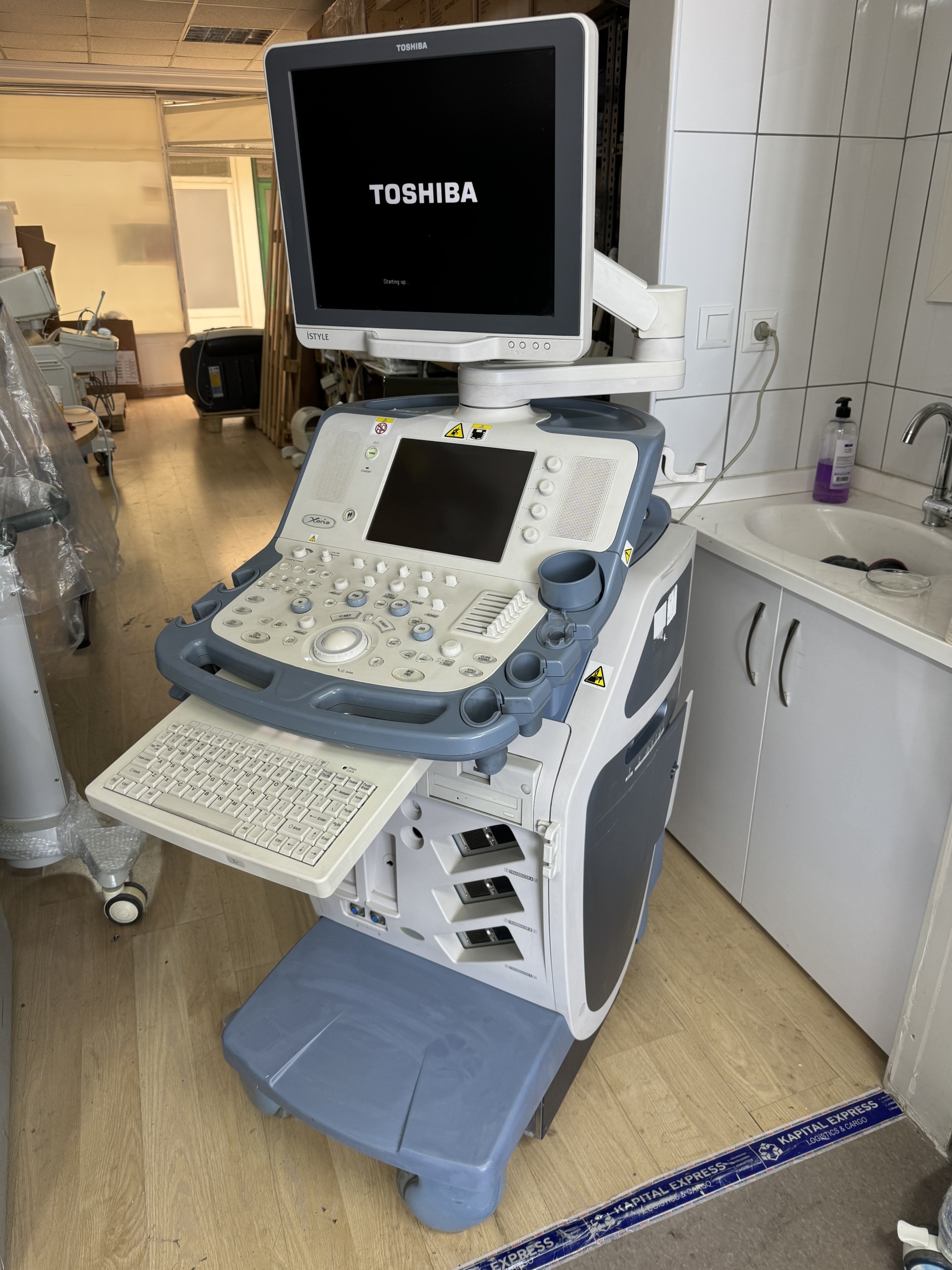
The images show one TOSHIBA Xario SSA-660A model ultrasound system, along with an integrated monitor and keyboard. The device’s control panel and mobile stand with wheels are also visible in the images. Furthermore, the device labels indicate the presence of a TOSHIBA USTS-770A model probe and a TOSHIBA USDF-770A model dynamic flow kit.
Brand and Model Information
- Main Device Brand-Model: TOSHIBA Xario SSA-660A
- Monitor Brand: TOSHIBA
- Accessory/Probe Brand-Model: TOSHIBA USTS-770A
- Accessory/Kit Brand-Model: TOSHIBA USDF-770A
Authenticity Assessment
Upon examining the general appearance of the device, the TOSHIBA logos, the quality of the labels, and the craftsmanship details, it is understood that the product is authentic. The “EXAMINED GOOD” and “QA” statements on the labels further support this conclusion.
Usage Areas
This device is a diagnostic ultrasound system. It can be used for a wide range of clinical applications, including abdominal, cardiac, transcranial, pediatric, and peripheral vascular examinations. Thanks to its various imaging modes, it enables precise visualization of tissue details and vascular structures. The TOSHIBA USTS-770A is an ultrasound probe used in conjunction with the device for imaging various body regions. The TOSHIBA USDF-770A, as a dynamic flow kit, enhances spatial resolution in color Doppler imaging, allowing for detailed examination of small vessels and their flow.
Quantity Information
The images show 1 TOSHIBA Xario SSA-660A ultrasound system, 1 TOSHIBA brand monitor, and 1 keyboard. According to the label information, there is 1 USTS-770A probe and 1 USDF-770A dynamic flow kit.
General Condition and Physical Deformation
The device is in used condition and is in good overall shape. No significant scratches, dents, cracks, or discoloration have been observed on the casing, screen, or other main sections. There may be slight surface dirt or dust on the control panel and keyboard.
Mechanical and Electronic Components
There are no visible issues with the mechanical components of the device, such as the wheels, hinges, buttons, and probe holders. The buttons on the control panel and the trackball appear to be intact. Regarding the electronic components, the “TOSHIBA” logo and “Starting up” text, along with “HDD 84% Free” information, are visible on the upper monitor, while menu and setting options are visible on the control panel screen. This indicates that the device is electronically functional.
Accessories
An integrated keyboard and probe holders are included with the device. No external cables or additional accessories are visible in the images. There are probe input sockets on the device. There is no information regarding the expiration dates of the accessories in the images.
Battery Status
There is no information in the images regarding the device’s internal or external battery, nor any details that would allow for a comment on the battery status.
Label Information
- Main Device (SSA-660A) Label Information:
- MODEL: SSA-660A – Indicates the model of the device.
- POWER: 230V~6.5A 50-60Hz 1500VA – Shows the electrical power requirements of the device.
- SERIAL No: 9909813172 – The unique serial number of the device.
- LEGAL MANUFACTURER: TOSHIBA MEDICAL SYSTEMS CORPORATION 1385, SHIMO-SHIGAMI, OTAWARA-SHI, TOCHIGI 324-8550, JAPAN – Contains legal manufacturer and address information.
- MEDICAL DEVICE CERTIFICATION No: 21600BZZ00530000 – Indicates the medical device certification number.
- EXAMINED GOOD QA – Indicates that the device has passed quality control and is in good condition.
- CE 0197 – The CE mark indicating that the device complies with European Union health, safety, and environmental protection standards.
- Accessory/Probe (USTS-770A) Label Information:
- MODEL: USTS-770A – Indicates the model of the probe.
- SERIAL NO: 140750246 – The unique serial number of the probe.
- LEGAL MANUFACTURER: TOSHIBA MEDICAL SYSTEMS CORPORATION 1385, SHIMO-SHIGAMI, OTAWARA-SHI, TOCHIGI 324-8550, JAPAN – Contains legal manufacturer and address information.
- Accessory/Kit (USDF-770A) Label Information:
- MODEL: USDF-770A – Indicates the model of the kit.
- SERIAL No: A4602012 – The unique serial number of the kit.
- LEGAL MANUFACTURER: TOSHIBA MEDICAL SYSTEMS CORPORATION 1385, SHIMO-SHIGAMI, OTAWARA-SHI, TOCHIGI 324-8550, JAPAN – Contains legal manufacturer and address information.
Screen Analysis
The “TOSHIBA” logo and “Starting up” text are visible on the upper monitor. Additionally, “HDD 84% Free” information is displayed on the screen. On the small screen on the control panel, various menu options, settings, and imaging modes (B, M, PW, CDI, 2D) are visible. This indicates that the device’s operating system is loaded and its basic functions are accessible.
Dimensions and Compatibility
There is no numerical information regarding the direct dimensions or size of the device in the images. However, the fact that it is a mobile system with wheels indicates that it can be easily transported in hospital and clinical environments and is suitable for use in different examination rooms. The device is of standard ultrasound system size.
Year of Manufacture
There is no direct information regarding the year of manufacture on the labels of the device or accessories in the images. The label format and the general design of the device suggest that it is a modern medical device, but there are insufficient visual clues for a concrete determination of the manufacturing year.
Documents
No invoices, warranty certificates, user manuals, or other documents belonging to the device are visible in the images.
Usage Duration
No information regarding the operating hours or total usage duration could be identified on the device screen, labels, or any documents present in the images.
Existing Faults
There is no clear evidence of any faults, such as broken parts, error messages, frayed cables, or missing components, anywhere on the device in the images. The fact that the device is on the startup screen indicates that its basic functionality is present.
Potential Risk of Failure
The general condition of the device appears to be good. No concrete situation indicating a potential risk of failure, such as severe wear, rust, or cable damage, has been observed in the images.