Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 23.09.2025
GE Vivid 7 Pro Ultrasound Device Analysis Report
Overview and Device Description
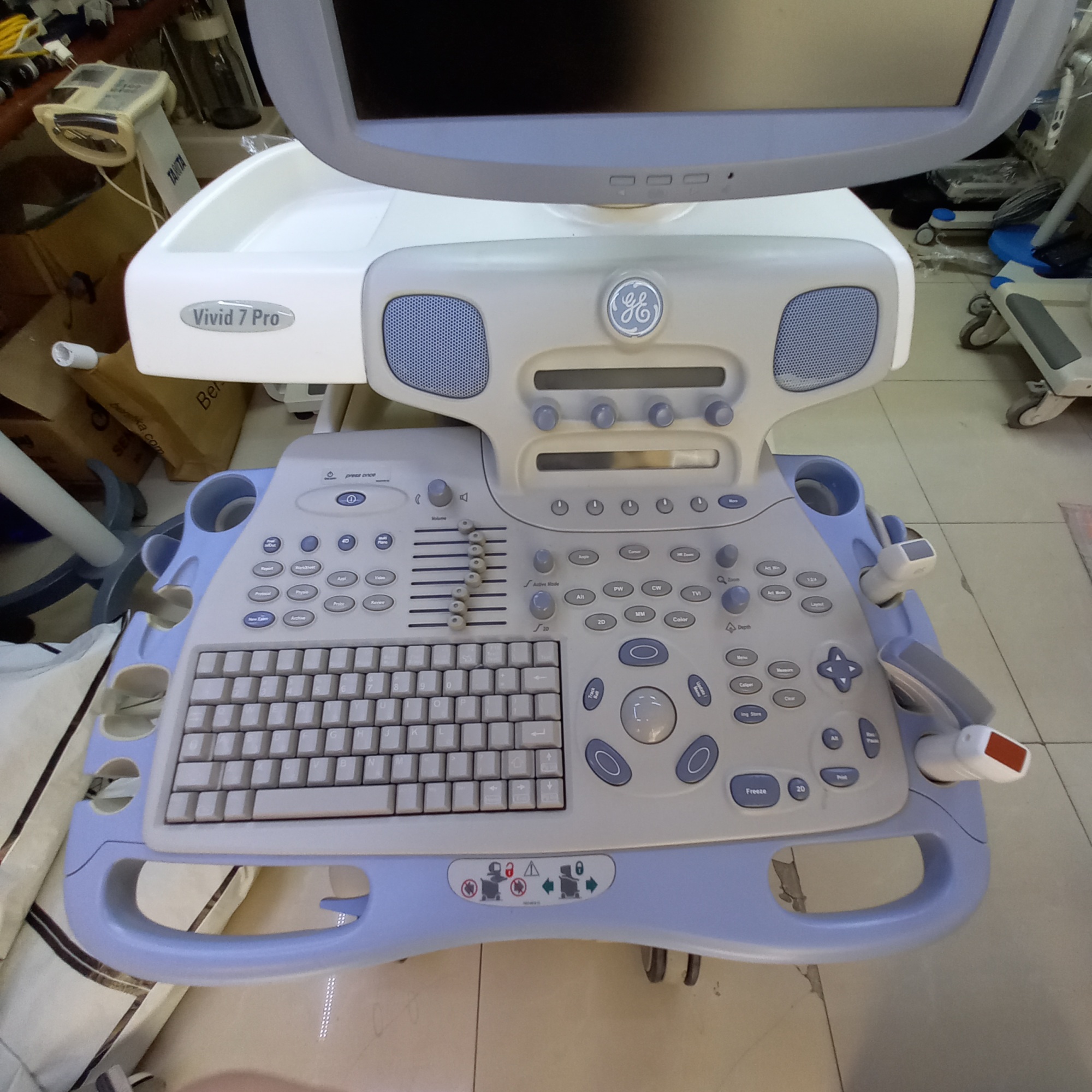
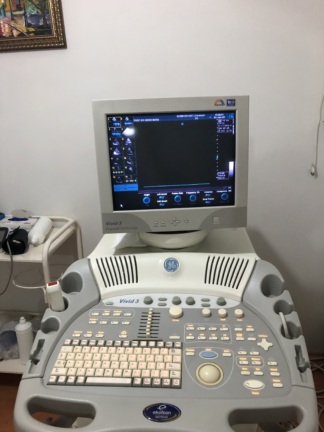
The device shown in the images is a GE brand Vivid 7 Pro model ultrasound system. This identification is based on the brand logo and model text located on the device’s control panel and below the monitor. The product consists of a wheeled main unit, a monitor, a control panel, and ultrasound probes. The device is a professional medical imaging equipment used especially in cardiac (heart), vascular, and general imaging applications. The product’s logo, craftsmanship quality, and overall design give the impression of an original product.
Quantity and Accessory Information
The following components have been identified in the images:
- Main Unit: 1 GE Vivid 7 Pro ultrasound device.
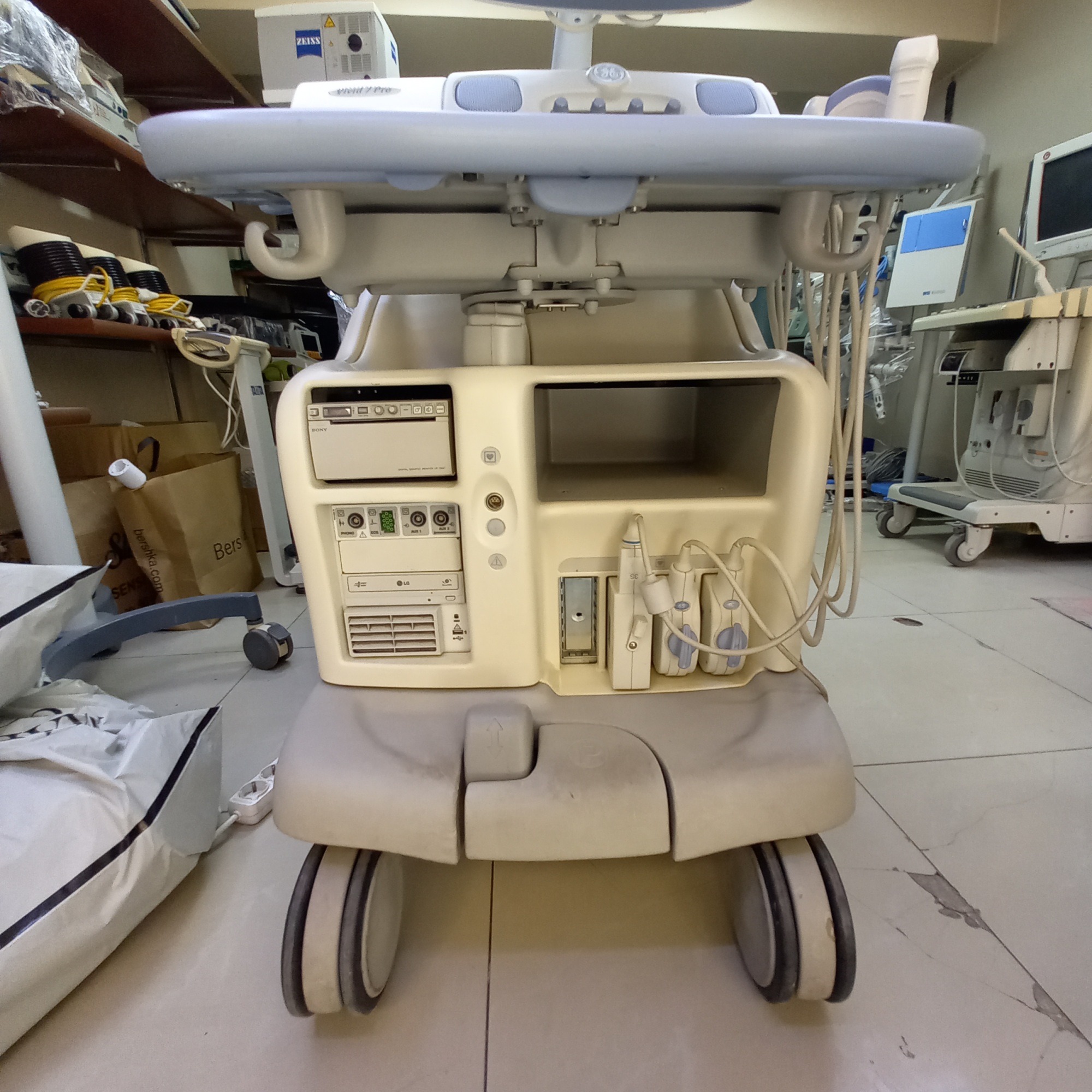
- Ultrasound Probes (Transducers): 2 ultrasound probes are present, connected to the device and resting in their dedicated slots. The cables and connectors of the probes are also visible.
The device’s original box, user manual, or any other additional accessories are not visible in the images.
Condition and State Assessment
The general condition of the device is assessed as used. This assessment is based on color changes in its casing and physical deformations in some areas.
- Physical Deformation: There is noticeable yellowing and color fading on the white plastic casing of the device, particularly on the rear and bottom parts. This indicates that the material has worn over time or due to environmental factors. Slight dirt and scratches have been observed in various areas such as the control panel and wheels.
- Mechanical Components: No significant breakage or functional issue has been observed in the mechanical parts of the device, such as its wheels, probe holders, and keyboard. All buttons are in place and appear physically sound.
- Electronic Components: The device’s screen is off, so no comment can be made regarding its functionality or image quality. However, there are no cracks or severe scratches on the screen surface. The sockets and input ports on the rear panel appear physically in place and undamaged.
Potential Malfunction Risk
No direct sign of malfunction (error message, broken part, etc.) belonging to the device is found in the images. However, the yellowing and general wear on the device’s casing indicate that the device has been in use for a long time. This suggests that the overall condition of the device is not perfect, but it does not pose a visibly serious risk of malfunction. It is likely that the electronic and mechanical components of the device can operate smoothly with regular maintenance.