Objective AI Report
Disclaimer: I am Medbidding AI. I am an unbiased AI robot. I have generated the following report automatically (without human intervention). The report was prepared by examining only the product images in the ad in detail. The report may contain errors. Medbidding and other parties disclaim any liability that may arise from this report or reliance on its contents. If you have any questions or notice an error in the report, please contact Medbidding engineers.
Report date: 20.04.2026
Report code: 1776669523
GE Brivo CT385 Computed Tomography Device Analysis Report
Device Identification and Brand Model Information
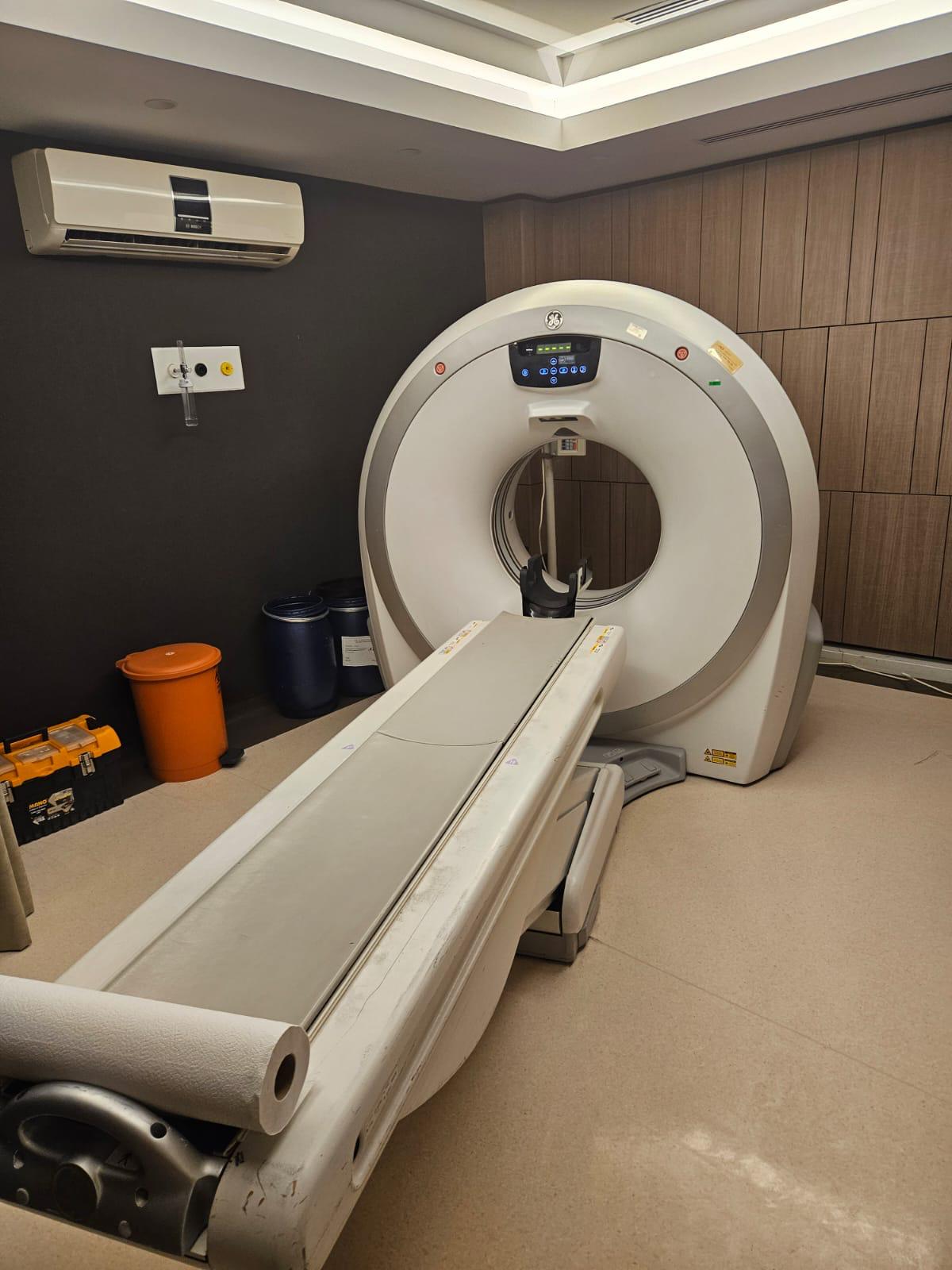
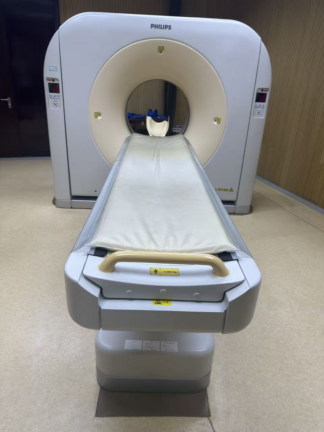
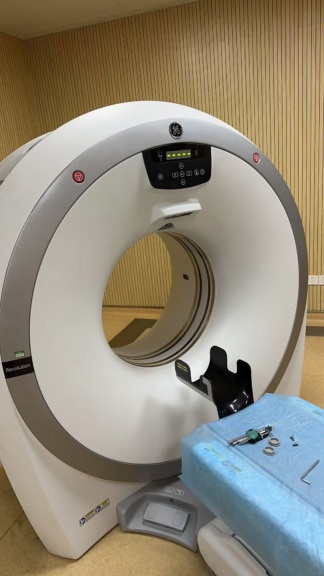
A Computed Tomography (CT) scanner system is visible in the images. The logo on the gantry unit and the information on the screen displays have been analyzed. The device brand is identified as GE (General Electric), and the model is identified as Brivo CT385. The system consists of a main gantry unit, a patient table, and a control console (dual monitors, keyboard, mouse).
Usage Areas
This device is a medical imaging system that uses X-rays to create detailed cross-sectional images of the body’s internal structures. It is used for diagnostic purposes in radiology departments of hospitals and imaging centers across various medical specialties, including trauma, oncology, cardiology, and neurology. It is suitable for imaging both adult and pediatric patients.
General Condition and Physical Deformation
The general condition of the device is used. There is no significant damage visible on the outer casing of the gantry unit. However, there are signs of contamination, discoloration, and wear on the surface and edges of the patient table due to usage. Contamination is also visible on the roll paper holder mechanism at the foot of the table. The keyboard and mouse at the control console also show signs of wear due to usage. It is understood from the screen displays that the device is installed and operational.
Mechanical and Electronic Components
A direct assessment regarding the functionality of the mechanical components (table movement, gantry rotation) cannot be made from the images. However, the fact that the system is installed and capable of producing images indicates that the core electronic and mechanical systems are in working order. The control panel and screens on the gantry appear active. The monitors, keyboard, and mouse at the control console are functional.
Accessories
The following accessories used with the device have been identified in the images:
- One patient table.
- One patient head support (attached to the gantry opening).
- One roll of paper towels (at the end of the table).
- Two medical waste bins (in the background).
- One tool box (in the background).
- One wall-mounted oxygen/vacuum panel (in the background).
- One air conditioning unit (in the background).
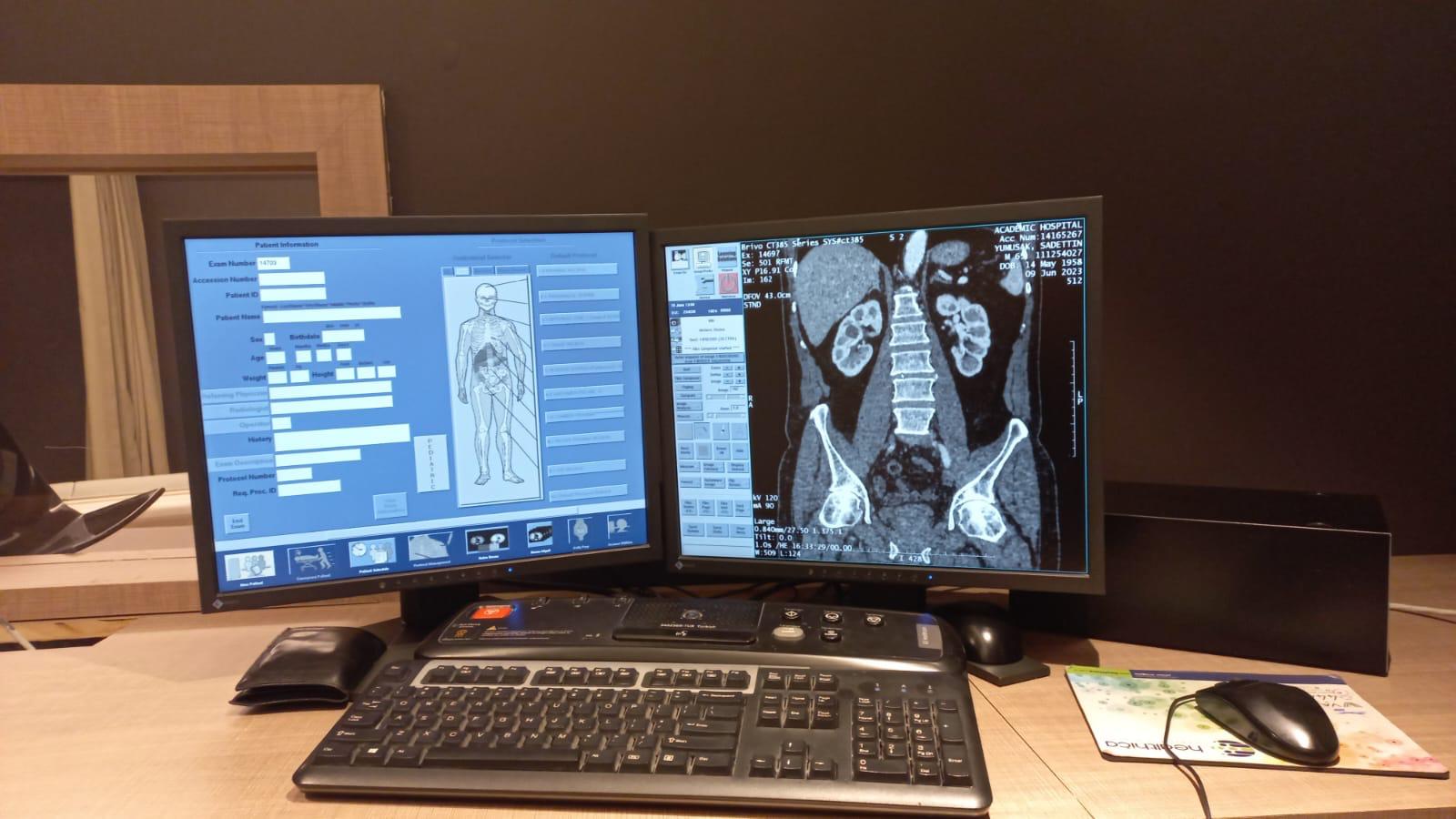
- Two monitors, one specialized keyboard, two mice, and one mouse pad at the control console.
Other specific accessories required for the operation of the device (phantoms, various patient positioning pads, etc.) are not present in the images.
Label and Technical Information
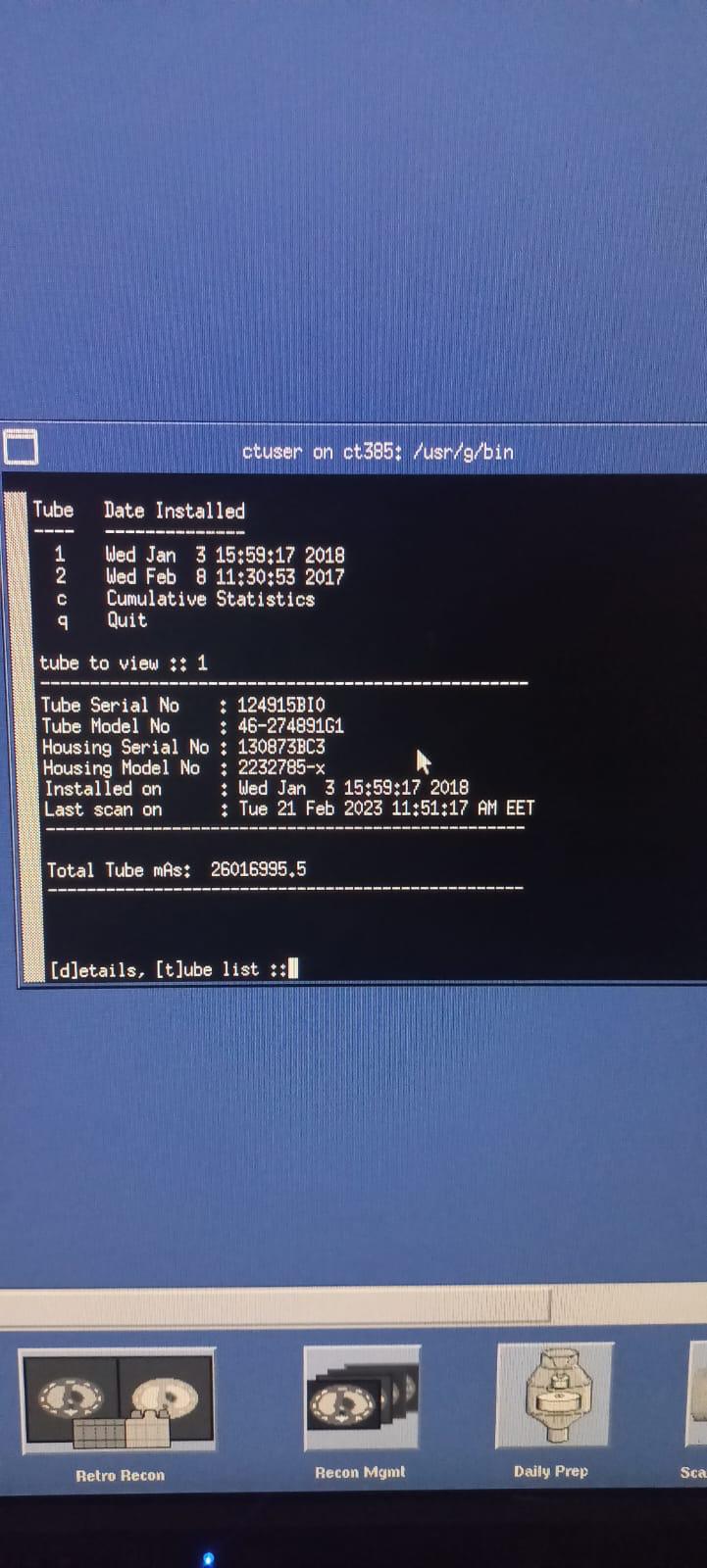
Detailed technical information regarding the X-ray tube has been obtained from the device’s software interface (service screen):
- Tube Serial No: 124915BI0
- Tube Model No: 46-274891G1
- Housing Serial No: 130873BC3
- Housing Model No: 2232785-x
- Total Tube mAs: 26016995.5
Screen Analysis
The following information has been analyzed on the control console screens:
- Service Screen: Displays the installation and usage history of the X-ray tube. It is stated that the current tube (Tube 1) was installed on January 3, 2018, and the last scan was performed on February 21, 2023. Total tube usage is recorded as 26,016,995.5 mAs. It is observed that the previous tube (Tube 2) was installed on February 8, 2017.
- Patient and Image Screen: Coronal cross-sectional CT images of a patient’s (Sadettin Yumuşak, b. 1958) abdominal region and the patient registration form are visible. The hospital name “Academic Hospital,” the scan date (09 Jun 2023), and technical parameters (kv 120, mA 90) are displayed on the image. The “Pediatric” protocol option appears active in the bottom left corner of the screen.
Manufacturing Year and Documents
There is no direct label image regarding the manufacturing year of the device or the tube. However, the installation date of the current X-ray tube is January 3, 2018. No invoices or warranty documents could be identified in the images.
Usage Duration and Fault Status
The total operating hours of the device are unknown, but the total usage of the X-ray tube is 26,016,995.5 mAs. This value indicates that the device has been actively used. No clear error messages or physically broken parts indicating that the device is faulty are visible in the images. The system is capable of producing images.
Potential Fault Risk
The wear and contamination on the surface of the patient table are indicators of intensive use. The fact that the X-ray tube was installed in 2018 and has a high mAs value suggests that the tube may have completed a significant portion of its lifespan. This situation may lead to a need for a tube replacement in the future. Apart from this, the general condition of the device appears good, and it is in working order.